Initial Clinical Assessment Before presenting in premature labor, many women experience one or more
of the following: spotting, an increase in vaginal discharge, and crampy
lower abdominal pain or pressure.49 The presence of these symptoms should be sought during antenatal visits, and
any woman complaining of these symptoms should be evaluated for
uterine contractions and cervical change. Once a patient presents with
painful regular uterine contractions, prompt assessment is necessary
because the success of all tocolytic therapy depends on therapeutic intervention
before uterine contractions have caused advanced cervical
dilatation.50, 51 Management of the patient should be dictated by the initial cervical examination. The
patient whose cervix is found to be long and closed should
be observed over time to assess whether or not contractions continue
and produce cervical change. However, the patient presenting with
a cervix 3 cm dilated and 50% effaced with regular uterine contractions
has a low therapeutic success rate and is best managed by administration
of tocolytic therapy as soon as possible. The patient should be placed
at bed rest in the left lateral decubitus position; she should undergo
fetal and tocodynometric monitoring, intravenous hydration, and
serial cervical examinations, preferably by the same examiner. This observation
period allows the clinician to distinguish between the 40% to 50% of
preterm labor patients who can be managed with bed rest and
intravenous hydration alone and who do not progress to delivery or need
pharmacologic therapy from the patients who are in true premature labor. Moreover, for
patients less than 3 cm dilated, it has not been shown
that waiting for cervical change alters the outcome of tocolytic therapy.33 During this observation period, the patient also should undergo a complete
medical and obstetric evaluation because prolongation of pregnancy
with tocolytic therapy may be contraindicated. In some patients, pregnancy
should not be continued for various obstetric and medical reasons (Table 2), and some clinical situations involve balancing the relative risk of
continuing the pregnancy with tocolytic therapy against the risk of preterm
delivery. Assessment should include a complete medical and obstetric
history and a full physical examination. Ideally, accurate assessment
of gestational age of the fetus will be accomplished in early pregnancy, but
because adequate prenatal care is neither available to nor
sought by all women, many preterm labor patients need ultrasound fetal
age assessment. Mindful of the error in fetal age assessment in late
pregnancy, many clinicians advocate fetal pulmonary maturity assessment
by amniocentesis either before initiating tocolytic therapy or shortly
after therapy begins. TABLE 2. Contraindications to Preterm Labor Inhibition
Absolute | Relative |
Fetal malformation incompat- | Placenta previa without life- |
ible with life | threatening bleeding |
Placental abruption | Chronic hypertension |
Fetal death | Fetal distress |
Severe pregnancy-induced | Mild fetal growth retardation Cervix |
hypertension or eclampsia | dilated more than 4 |
Placenta previa with life- | cm in a singleton preg- |
threatening bleeding | nancy or more than 5 cm |
Chorioamnionitis | in a multiple gestation |
Severe fetal growth retardation | |
Any maternal medical condi- | |
tion in which prolongation | |
of the pregnancy is unwarranted | | An underlying infectious etiology for preterm labor must be sought so that
diagnosis may be established and appropriate antibiotic therapy initiated. The
association of preterm labor with urinary tract infections
necessitates a urine examination by both microscope and culture. A meta-analysis
has revealed that antibiotic treatment significantly reduces
the risk of delivery of a low-birth-weight infant in this population.52 Pyrexia associated with infection should be treated because an elevated
maternal temperature may itself lead to uterine contractions.53 Cervical cultures for gonorrhea, group B streptococcus, and chlamydia should be obtained and are useful in patient management. Of particular concern are signs and symptoms of chorioamnionitis, a cause of preterm labor that may be present with intact membranes with
no other signs or symptoms except preterm labor.54 Routinely available tests for subclinical chorioamnionitis include maternal
serum C-reactive protein, a Gram's stain of unspun amniotic
fluid, a white blood cell count, and an amniotic fluid glucose concentration. Although
C-reactive protein has a low specificity and sensitivity
for intra-amniotic infection, its high negative predictive value
allows it to exclude amniotic infection as a cause for preterm labor.55 A Gram's stain has a low sensitivity, but it is highly specific for
intra-amniotic infection and has a high positive and negative predictive
value.35 A study of white blood cell counts of amniotic fluid obtained from preterm
labor patients has shown that cell counts greater than 50 cells/mm3 have a high sensitivity for intra-amniotic infection, but a low specificity.37 In one study, amniotic fluid glucose concentrations were lower among patients
with intra-amniotic infections; amniotic fluid glucose concentrations
less than 14 mg/dL had a high sensitivity and specificity for
intra-amniotic infections, but had a low positive predictive value.38 Because no one test has been demonstrated to have high specificity and
sensitivity with high positive and negative predictive values, combinations
of these clinical tests seem appropriate. Patients whose uterine contractions do not respond to hydration and decreased
activity, and whose contractions cause observed cervical change, and who have no contraindications to pregnancy prolongation or to the use
of tocolytic agents are candidates for tocolytic therapy. Pharmacologic Interventions After the decision to inhibit preterm labor is made, the physician must
decide which of the available therapeutic modalities to use. Historically, the
obstetrician has used narcotics (e.g., morphine, meperidine) and sedatives as tocolytic agents; however, these
agents do not cause smooth muscle relaxation56, 57, 58, 59 and really have no place in the therapy of true preterm labor. Intravenous
ethanol was used as a tocolytic agent during the 1960s and 1970s, but
it has been abandoned because of maternal inebriation and a higher
incidence of adverse neonatal outcome.60, 61 Current pharmacologic therapies for preterm labor include the β-sympathomimetics, magnesium
sulfate, prostaglandin synthetase inhibitors, slow
calcium-channel antagonists, and as a research agent, the oxytocin
receptor antagonist atosiban. No agent has been found to be significantly
better than all others, and in any individual clinical situation
one or another of these agents may be preferable. b-SYMPATHOMIMETICS. Pharmacology. In 1925, Rucker62 discovered that small doses of epinephrine inhibited uterine contractions. It
was not until 1948, however, that Ahlquist63 demonstrated that this effect was caused by stimulation of β-receptors
in uterine smooth muscle. Lands and colleagues64 further showed that the β-adrenergic receptors could be divided into
two distinct groups: the β1-receptors, found in cardiac muscle, the small intestine, and adipose tissue; and
the β2-receptors, found in the smooth muscle of the uterus, blood vessels, bronchioles, and
diaphragm. Stimulation of β1-receptors has positive inotropic and chronotropic effects on heart muscle
and causes relaxation of intestinal smooth muscle, lipolysis, and
glycogenolysis. Stimulation of β2-receptors relaxes the smooth muscle of the uterus, bronchioles, and blood
vessels. Although drugs are classified as having predominantly β1 or β2 effects, the difference between β1- and β2-receptors is not as distinct as that found between α- and β-receptors, and
there is considerable cross-reactivity between drugs classified
predominantly as a β1-agonist or a β2-agonist.65 A drug classified as a β2-agonist may lose its β2-specificity at higher doses. The β-sympathomimetics are all derived from structural modifications
of the epinephrine molecule. Increasing the size of the alkyl substituent
at the amino group increases β-agonist activity, and substituting
a hydroxyl group at the 3 and 5 positions on the aromatic ring
confers greater β2-receptor specificity.65 Drugs whose design has been based on these structure-activity relationships
and that have been shown to have greater β2-receptor agonist activity are fenoterol, terbutaline, ritodrine, salbutamol, and
hexoprenaline. Clinical studies have demonstrated that all
these drugs have tocolytic efficacy,66, 67, 68, 69, 70 but in the United States, ritodrine, the only drug approved by the FDA
for preterm labor therapy, and terbutaline are the β2-sympathomimetics commonly used. The β2-sympathomimetics cause smooth muscle relaxation by interaction with the β-receptor
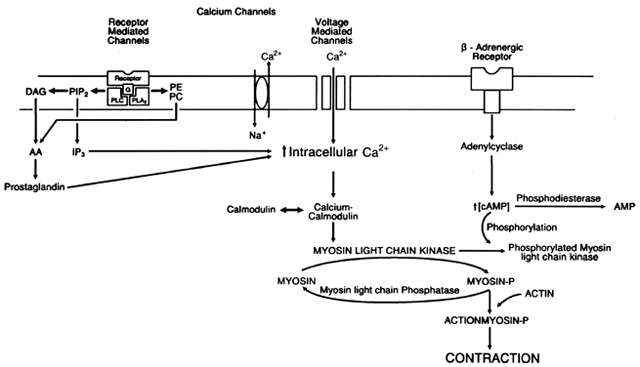
on the cell membrane. This agonist-receptor complex, through
stimulation of a GTP-binding protein activates adenyl cyclase, which
converts ATP to cAMP.71 Increasing intracellular cAMP activates cAMP-dependent protein kinases
that cause phosphorylation of a protein associated with the sodium-potassium
ATPase system. This in turn increases the rate at which Na+ is pumped out of the cell in exchange for K+. An increase in the Na+ gradient across the cell membrane accelerates the Na+-Ca2+ exchange, causing a net loss of Ca2+ from the cell. The decrease in intracellular calcium deactivates myosin
light-chain kinase and causes smooth muscle relaxation. An increase
in intracellular cAMP also inhibits myosin light-chain kinase activity
directly by causing phosphorylation of the enzyme itself and further
decreasing intracellular calcium by promoting Ca2+ binding by the sarcoplasmic reticulum. By altering adenyl cyclase activity
and the calcium pump, β-adrenergic agonists control the activity
of myosin light-chain kinase, the key enzyme controlling the interaction
of actin and myosin, thereby influencing the basal contractile
state of uterine smooth muscle.72, 73, 74 Maternal, Fetal, and Neonatal Effects. β2-sympathomimetics produce a dose-related increase in maternal heart rate
and a widening of the pulse pressure.75 Tachycardia is caused by both direct stimulation of cardiac β1-receptors and a reflex baroreceptor activation due to peripheral vasodilation. The
lowered diastolic pressure may facilitate venous return to
the heart, thereby increasing stroke volume and systolic pressure. Cardiac
output has been shown to increase by as much as 56% when β-adrenergic
agonists are used.75 This is of concern because these drugs are used at a time during pregnancy
when physiologic changes already have increased cardiac output by
approximately 40% over prepregnancy levels. Maternal metabolic effects caused by the use of β2-sympathomimetics can be traced to the increased intracellular cAMP and
include hyperglycemia from hepatic glycogenolysis, hyperlacticacidemia, lipolysis, and
hyperinsulinemia caused by a direct effect of the β-agonist
on the pancreatic β-cells.76, 77, 78, 79, 80, 81 Because β-sympathomimetics act on the sodium-potassium pump, they
also reduce the plasma K+ concentration by shifting K+ from the extracellular space to the intracellular space.82, 83 The hyperglycemia associated with β-sympathomimetics used for tocolysis
is seen mainly during intravenous administration of the drugs. The
highest glucose concentrations are seen approximately 3 hours after
intravenous therapy is initiated, begin to decline thereafter, and return
to normal after therapy is stopped.83 Continued oral administration of ritodrine does not appear to affect carbohydrate
metabolism adversely,84, 85 but oral terbutaline has been shown to impair maternal glucose tolerance
and to cause the development of gestational diabetes.86 The hypokalemia associated with β-sympathomimetics is seen only during
intravenous therapy, and K+ concentrations begin to return to normal during the first 24 hours of
therapy.83 The β-sympathomimetics also affect total body water by stimulating
the renin-aldosterone system and the release of antidiuretic hormone, resulting
in a net increase of total body sodium and water.87, 88, 89 A study of pregnant baboons revealed that this increase in total body
water results in an increase in the interstitial fluid space and does
not increase plasma volume.90 The use of β-sympathomimetics is associated with the common side effects
of nervousness, restlessness, anxiety, tremors, palpitations, nausea, and
vomiting, which are caused by sympathomimetic stimulation. The
hyperglycemia associated with β-sympathomimetics has precipitated
diabetic ketoacidosis when used in diabetics in whom blood sugars
were not monitored.91, 92, 93 However, the most serious adverse reactions seen with the β-sympathomimetics
used for tocolysis relate to effects on the cardiovascular
system. A rare maternal death was reported among patients with unrecognized
cardiac or pulmonary disease.94 Cerebral ischemic episodes have been reported in patients with a history
of migraine headaches.95 Chest pain or tightness was reported among 1% to 2% of patients treated
with intravenous β-sympathomimetics.96 Electrocardiographic recordings also were found to exhibit ischemic ST-segment
depression on occasion,97 but measurements of cardiac enzymes during β-sympathomimetic therapy
have not revealed any evidence of myocardial damage.98, 99 Cardiac arrhythmias can occur in approximately 2% of patients during intravenous
therapy, are usually asymptomatic supraventricular arrhythmias, and
respond to discontinuation of therapy.100, 101, 102 Dyspnea occurs in 5% to 12% of patients treated with intravenous β-sympathomimetic
agents and may reflect pulmonary congestion.103 During the first 24 to 48 hours of therapy, the prevalence of frank pulmonary
edema has been reported to occur in 0.3% to 5% of patients.104, 105 Patients with multiple gestations who have even higher blood volumes than
those with singleton gestations seem particularly vulnerable to the
occurrence of pulmonary edema. Originally, glucocorticoids were thought
to increase the risk for pulmonary edema, but this observation has
not been corroborated in larger studies.102 The pathophysiology of the pulmonary edema seen with β-sympathomimetic
use is poorly understood. Left ventricular failure associated with
iatrogenic fluid overload, prolonged exposure to a high dose of a sympathomimetic
drug, and increased pulmonary capillary permeability coupled
with lowered intravascular colloid osmotic pressure are tentative
explanations. Hemodynamic monitoring of a few such patients with Swan-Ganz
catheters has revealed that this pulmonary edema may not represent
congestive heart failure because pulmonary wedge pressures have been
found to be normal.106 This lends support to the hypothesis that the mechanism of action is noncardiac
pulmonary edema caused by altered pulmonary capillary permeability, similar
to the pulmonary edema caused by epinephrine and norepinephrine. However, a
controlled experimental study of ritodrine in baboons
did not reveal pulmonary capillary membrane changes.107 In another study, noninvasive techniques in humans revealed a progressive
elevation in pulmonary wedge pressures, suggesting that left ventricular
failure is the pathophysiologic cause of the pulmonary edema.108 Fetal effects of β-sympathomimetics can be traced to placental transfer
of the active drug or to altered maternal status. Both terbutaline
and ritodrine cross the placenta, with ritodrine passing more readily.109 During intravenous therapy with β-sympathomimetics, a mild fetal
tachycardia and increased fetal heart rate variability can be seen.110, 111 Fetal breathing movements also increase with terbutaline therapy.112 Infants whose mothers were treated with subcutaneous terbutaline within 2 days
of delivery can develop transient hypoglycemia associated with
elevated cord insulin levels.113 Fetal cardiac effects reported include myocardial necrosis associated
with prolonged antenatal use of terbutaline,114 supraventricular tachyarrhythmias causing fetal hydrops,115, 116 and ventricular septal thickening.117 In prospective controlled studies of ritodrine, however, these effects
on the neonate have not been seen. A large-scale retrospective analysis
controlling for many confounding variables demonstrated an association
between maternal use of a β-mimetic agent for preterm labor tocolysis
and the occurrence of neonatal periventricular-intraventricular
hemorrhage.118 Use of a β-mimetic was associated with more than a twofold increase
in risk of neonatal periventricular-intraventricular hemorrhage, with
the same increase in risk for the occurrence of grades 3 and 4 periventricular-intraventricular hemorrhage. Nevertheless, a neonatal outcome
study and follow-up studies from 1 to 9 years of infants born to women
treated with β-sympathomimetics have found no demonstrable abnormalities
in condition at birth or overall growth and development.119, 120 Usage Guidelines. After a patient has been assessed for any contraindications to preterm
labor inhibition, use of a β-sympathomimetic requires that the patient
be assessed for any absolute or relative contraindications specific
to their use (Table 3). Baseline maternal vital signs, a 20- to 30-minute fetal monitor strip, blood
chemistries, and an electrocardiogram should be obtained before
therapy is begun. Treatment with ritodrine initially is begun by intravenous
administration with a controlled infusion device. Terbutaline
has been shown to have unacceptable cardiovascular side effects when
administered intravenously, and this mode of administration has been
abandoned.121 Subcutaneous administration of terbutaline has been reported, however, with
minimal maternal and fetal side effects.122 TABLE 3. Recommended Contraindications to Use of β—Μimetic
Drugs
Absolute | Relative |
Gestational age <20 weeks | Placenta previa without life- |
or > 36 weeks | threatening bleeding |
Maternal cardiac disease | Cardiac disease (Class I) |
(Class II-IV) | Diabetes |
Thyrotoxicosis with active | History of migraine head- |
disease | aches |
Moderate to severe pre- | Benign cardiac arrhythmias |
eclampsia or eclampsia | |
Placental abruption | |
Placental previa with life- | |
threatening bleeding | |
Maternal condition in which | |
increased maternal cardiac | |
output may be hazardous | | Used for uterine tocolysis, ritodrine should be begun only after initial
intravenous hydration because the β-sympathomimetics cause peripheral
vasodilation and can cause hypotension. A urine-specific gravity
test can be used as a rough assessment of hydration status in a given
patient. A urine-specific gravity greater than 1.020 would dictate a
greater fluid need, and 1000 mL should be infused over a period of 30 to 60 minutes. A
more dilute urine, with a specific gravity less than 1.020, would
dictate less fluid need, and 500 mL would be infused over
the same time period. Ritodrine is administered diluted in normal saline at an initial dose of 0.05 mg/minute. The
dose is increased at 10- to 30-minute intervals
until contractions cease or are significantly reduced or until unacceptable
side effects develop. Before the infusion rate is increased, the
maternal pulse and blood pressure should be monitored, and uterine activity
and fetal heart rate should be noted. Because β-sympathomimetics
cause salt and water retention, the mainline intravenous infusion
rate should be adjusted with each increase in dose so that total intravenous
fluids do not exceed 100 to 120 mL/h or 2500 mL during a 24 hour
period. Tocolytic therapy with β-sympathomimetics is titrated
not only to suppress uterine activity, but also to maintain maternal
pulse rate and blood pressure within acceptable limits. The maternal
pulse should increase by approximately 20 to 40 beats/minute over the
baseline pulse rate. If no maternal pulse increase is noted, the drug
dose can be increased until contractions cease, even if recommended maximum
doses are exceeded. The dose should not be increased, however, if
maternal pulse rate is 130 beats/minute or more. A widening of the pulse
pressure also can be expected, which can cause a significant decrease
in blood pressure. These cardiovascular changes can be reversed by
a decrease in the ritodrine infusion rate or by an increase in intravenous
fluids. Care must be exercised, however, not to overload the patient
with fluids. Once uterine tocolysis is achieved, the infusion is continued for 6 to 24 hours. The
manufacturer recommends that the ritodrine infusion be continued
at the infusion rate at which uterine relaxation occurred.123 Caritis and colleagues124 made an alternative recommendation. Because the manufacturer's regimen
can result in excessively high plasma concentrations of ritodrine
and may be related to adverse maternal cardiovascular side effects, they
suggest that, after uterine tocolysis has been achieved, the ritodrine
infusion rate be reduced to the lowest infusion rate that maintains
continued uterine relaxation and that the infusion rate be continued
for 12 hours. An alternative regimen to the use of intravenous ritodrine has been described
in which a dose of 0.25 mg terbutaline is administered subcutaneously
and repeated hourly, if maternal pulse rate is less than 120 beats/minute, until
uterine contractions cease.122 Terbutaline is then continued orally in a dose of 5 to 10 mg every 4 hours. Efficacy
in a retrospective review was found to be comparable to
the efficacy of ritodrine. Subcutaneous terbutaline also has been used
to decrease myometrial activity and to increase uteroplacental blood
flow in the therapy of fetal distress.125, 126 Oral medication is begun 30 minutes before the intravenous infusion is
discontinued. Oral therapy should be titrated to sustain an increase in
maternal pulse rate, which should remain between 100 and 120 beats/minute, or 20 to 40 beats/minute over the baseline maternal heart rate. A
comparison of oral ritodrine and terbutaline indicates that terbutaline
may be more effective in preventing recurrent labor.127 The need for continued oral therapy, however, has been questioned by many. The
original studies that showed a decrease in the incidence of recurrent
episodes of preterm labor used uterine contractions, not cervical
change, as the criterion for preterm labor.128 When stricter criteria for establishing the diagnosis of preterm labor
were used in a randomized clinical trial, oral terbutaline was not shown
to reduce the rate of preterm birth.129 If uterine contractions recur, the maternal pulse rate will indicate whether
a supplemental oral dose can be given safely in an attempt to restore
tocolysis without resorting to intravenous therapy. With continued
contractions and demonstrated cervical change, intravenous therapy
can be reinstituted if indicated in the overall management of the preterm
labor patient. Clinical Efficacy. Evidence of the clinical efficacy of the β-sympathomimetics in the
therapy of preterm labor has been provided by prospective clinical trials
of ritodrine and terbutaline, which have demonstrated their ability
to suppress uterine contractions and delay delivery.41, 42, 50, 130 Initial trials, however, used varying protocols of administration at different
centers, an inadequate number of controls, and nonstandardized
treatments in the control population. Subsequent randomized trials of
ritodrine and terbutaline, both of which seem to have equal efficacy
in arresting preterm labor, have failed to show a benefit of these β-sympathomimetics
over placebo in the long-term prolongation of pregnancy.104, 131, 132, 133, 134, 135, 136 Critical assessment, including a meta-analysis of existing clinical trials
of the β-sympathomimetics, has shown that β-sympathomimetics
can delay delivery for at most 24 to 48 hours and that the drugs
have no effect on perinatal morbidity or mortality.137 The clinical value of the β-mimetics, however, may be in that they
provide a delay in delivery and allow administration of corticosteroids
and transfer to a tertiary care center for delivery. Use of the β-sympathomimetics in patients with premature rupture of
the membranes has been studied in three prospective randomized studies.138, 139, 140 The overall weight of the clinical evidence is that their use resulted
in no significant difference in delay of delivery or neonatal outcome
between the treated group and the control group, and their use was therefore
of no benefit. MAGNESIUM SULFATE. Pharmacology. Essential as a cofactor for many cellular enzyme systems and in neuromuscular
transmission, magnesium is a naturally occurring intracellular
cation. In the 1950s Hall and colleagues141 observed that elevated levels of Mg2+ reduced uterine contractility in vitro and prolonged labors in patients
treated with magnesium sulfate for preeclampsia. Magnesium sulfate alters
the extracellular and intracellular Mg2+ concentration, which in turn inhibits Ca2+ entry through calcium channels into the myometrial cell.142, 143 Magnesium sulfate does not appear to effect Ca2+ release from intracellular stores. The net effect of magnesium sulfate
on uterine smooth muscle is a decrease in intracellular Ca2+ and inhibition of myosin light-chain kinase and the binding of actin and
myosin. The normal plasma concentration of Mg2+ is 1.5 to 2.2 mEq/L. Mg2+ is eliminated by glomerular filtration through the kidney and is reabsorbed
in the proximal renal tubules.144 Serum Mg2+ concentrations have been reported to decrease during early pregnancy, to
increase gradually throughout the second and third trimesters, and
to decrease again immediately before delivery.145 A high plasma Mg2+ concentration is toxic and is rarely seen in persons with normal renal
function. Elevated levels of Mg2+ depress neuromuscular function by inhibiting acetylcholine release at
the motor nerve end plate. Clinically this is manifested by loss of deep
tendon reflexes. Although there have been reports of loss of deep tendon
reflexes at Mg2+ plasma levels as low as 4 mEq/L, generally deep tendon reflexes are not
lost until plasma concentrations reach levels of 8 to 10 mEq/L.146 Respiratory depression occurs with plasma concentrations of 12 to 15 mEq/L.146, 147 At plasma levels of 10 to 15 mEq/L, Mg2+ prolongs cardiac conduction time as well as the PR and QRS intervals. Plasma
concentrations greater than 15 mEq/L cause cardiac arrest.148 Maternal, Fetal, and Neonatal Effects. Common maternal side effects of magnesium sulfate therapy include flushing, a
warm sensation, headache, lethargy, dizziness, nausea, and blurred
vision. Cardiovascular effects include a decrease in peripheral vascular
resistance, a small increase in cardiac output, a mild increase
in heart rate, and a widening of the pulse pressure.148 From animal studies, magnesium sulfate has been shown to produce a mild
increase in uteroplacental blood flow and to maintain perfusion pressure.149, 150 Pulmonary edema has been reported in approximately 1% of patients treated
with magnesium sulfate therapy for preterm labor.151 During magnesium sulfate therapy, maternal Ca2+ levels decrease by approximately 25%, which is caused by increased urinary
Ca2+ excretion.152, 153 Maternal bone density has been shown to decrease in magnesium sulfate-treated
patients compared with controls.154 Distributed through total body water, Mg2+ readily crosses the placenta, with fetal and neonatal levels proportional
to maternal levels.152 Mg2+ has been shown not to affect fetal heart rate variability,155, 156 but it may cause a nonreactive nonstress test and a decrease in fetal
breathing movements.157 Immediate neonatal effects include respiratory depression and decreased
muscular tone; generally one to two Apgar points are deducted for decreased
tone and drowsiness.158 These adverse effects most likely are related to the Mg2+ umbilical cord level at delivery and the duration of magnesium sulfate
therapy.159, 160, 161, 162, 163 Prolonged magnesium sulfate infusions generally begun during the second
trimester of pregnancy have been shown to cause demineralization of
the fetal long bones, rachitic bony abnormalities, and dental enamel hypoplasia.164 Bony abnormalities can resolve during the neonatal period, but dental
enamel hypoplasia may persist.165 Usage Guidelines. Used as a tocolytic agent, the empiric therapeutic range of Mg2+ is 4 to 8 mEq/L,158, 166 which is achieved after a loading dose of 4 to 6 g of magnesium sulfate
infused for a 20 minute period, followed by infusion rates of 1 to 3 g
of magnesium sulfate every hour, but higher infusion rates may be necessary. Tocolysis
with magnesium sulfate has not always been correlated
with serum levels, and barring clinical indication of toxicity, higher
infusion rates have been reported.167 Total parenteral fluids should be controlled strictly during therapy because
overhydration is the mechanism by which magnesium sulfate causes
pulmonary edema.168 Monitoring of the patient during therapy includes periodically checking
for deep tendon reflexes, monitoring urine output, and measuring plasma
Mg2+ concentrations, especially in patients with altered renal function or
decreased urinary output. Usually magnesium sulfate is infused for 12 to 24 hours, but
some patients require longer periods of treatment. Because
magnesium sulfate is not tolerated orally, many clinicians use oral β-sympathomimetics for long-term management. Alternatively, magnesium
gluconate or oxide, magnesium salts with fewer gastrointestinal
side effects, can be given in a dose of 1 g every 2 to 4 hours and
are reportedly as effective as ritodrine with less adverse side effects.169, 170, 171 Clinical Efficacy. The clinical efficacy of magnesium sulfate first was reported in an observational
study by Spisso and co-workers,172 which showed that magnesium sulfate can delay delivery in preterm labor
patients with intact membranes for 48 hours in 71% of patients, for
more than 7 days in 45% of patients, and until term in 31% of patients. Elliott,173 reporting on a retrospective analysis of preterm labor patients treated
with magnesium sulfate, found that the greatest success could be achieved
in singleton pregnancies with intact membranes and that therapeutic
success was directly related to the degree of cervical dilatation
on admission. Labor was arrested for 48 hours in 87% of patients with
cervical dilatation of 2 cm or less, in 62% with dilatation of 3 to 5 cm, and
in 31% with dilatation of 6 cm or more. The efficacy of magnesium sulfate was first shown in a randomized comparison
trial using intravenous ethanol and dextrose as control therapies.43 This study indicated that magnesium sulfate was more effective than alcohol
in inhibiting uterine contractions for 24 hours, and therapeutic
success was correlated with the degree of cervical dilatation at the
time treatment was begun. Cotton and associates134 reported on a clinical trial randomizing preterm labor patients to magnesium
sulfate therapy, terbutaline therapy, or placebo; this study found
no difference among these modalities in their ability to delay delivery
for 48 hours. The largest clinical trial included 156 women randomized
either to magnesium sulfate or to placebo.174 Criteria for admission into the study did not include cervical change
under observation, and 42% of the treated group and 64% of the control
group delivered more than 1 week after treatment. Magnesium sulfate was
found to have no significant effect on duration of gestation, birth
weight, neonatal morbidity, or perinatal mortality. Mean delay of delivery
for the treated group was 26 hours versus 22 hours for the control
group. Reports from clinical trials comparing magnesium sulfate to β-sympathomimetics
have shown that magnesium sulfate is as effective
as the β-sympathomimetics in suppressing preterm labor, but magnesium
sulfate has fewer maternal and fetal side effects.121, 175, 176, 177, 178 These findings have prompted the suggestion that magnesium sulfate be
used as the primary tocolytic agent and that ritodrine be reserved for
patients whose contractions cannot be inhibited with magnesium sulfate. Magnesium
sulfate is also probably safer for use if fetal status is
at all compromised because it does not impair uteroplacental perfusion.150 In attempts to improve clinical efficacy, magnesium sulfate has been combined
with ritodrine in patients whose labors could not be arrested with
ritodrine alone. In one randomized clinical trial of combined magnesium
sulfate and ritodrine compared with magnesium sulfate alone, combined
therapies produced mild improvement in tocolytic efficacy but were
associated with a greater number of cardiovascular side effects.179 Hatjis and colleagues180 reported on combined therapy of ritodrine plus magnesium sulfate in a
highly select group of patients failing ritodrine tocolysis alone and
showed a 60% success rate, with no increase in maternal or fetal complications. In
a randomized clinical trial comparing the efficacy of ritodrine
alone to the combination of ritodrine plus magnesium sulfate, ritodrine
plus magnesium sulfate was found to be more efficacious than
ritodrine alone and did not appear to increase the frequency of adverse
side effects.181 The majority of clinical trials on magnesium sulfate therapy of preterm
labor therefore show that magnesium sulfate therapy has similar efficacy
to the β-sympathomimetics and at best delays delivery for 24 to 48 hours. PROSTAGLANDIN SYNTHETASE INHIBITORS. Pharmacology. Prostaglandins stimulate the intracellular influx of Ca2+ and cause release of Ca2+ from the sarcoplasmic reticulum.182, 183 Prostaglandins also enhance myometrial gap junction184 formation and may represent the final common biochemical mediator leading
to uterine contractions and labor. Nonsteroidal anti-inflammatory
agents, such as aspirin, indomethacin, ibuprofen, and naproxen, have been
shown to inhibit the enzyme cyclo-oxygenase, which converts arachidonic acid to prostaglandins in the first step of
the prostaglandin cascade, hence the name prostaglandin synthetase inhibitors. Of
all the anti-inflammatory agents, aspirin is the only one
that inhibits cyclo-oxygenase irreversibly.185 Of the available agents, indomethacin is the agent that has been most
studied as a tocolytic agent. Indomethacin is a potent inhibitor of cyclo-oxygenase, having anti-inflammatory, analgesic, and
antipyretic activity. Renal effects of prostaglandin
synthetase inhibitors include a reduction in glomerular filtration
rate, reduced sodium and water clearance, and lower plasma renin
activity, making their use inadvisable in patients with renal disease.185 Indomethacin has been reported to cause neutropenia, thrombocytopenia, and
in rare instances aplastic anemia. Platelet function is altered by
inhibition of platelet agglutination, causing a prolonged bleeding time, and
peripartum use has been associated with postpartum hemorrhage.186 Gastrointestinal side effects include anorexia, nausea, abdominal pain, and
ulcerative lesions of the gastric and intestinal mucosa. Rare, but
fatal, cases of hepatitis have been reported. The central nervous system
can be affected most commonly by severe frontal headaches, but dizziness, depression, and
psychosis have been seen with prolonged use
of indomethacin.185 Indomethacin, whether administered orally or rectally, is absorbed rapidly
and completely; peak plasma levels occur 1 to 2 hours after administration.187 Time to peak indomethacin serum concentration after oral administration
occurs less rapidly than after rectal administration, and peak serum
concentration can be further delayed by ingestion with food.187 To minimize gastrointestinal discomfort from the drug, indomethacin can
be administered with food. Peak plasma concentrations of indomethacin
given orally to laboring patients are reached somewhat later and are
lower than plasma concentrations found in nonpregnant women.188 Studies in nonpregnant adults show that the half-life has great biologic
variation, ranging from 2.6 to 11.6 hours, with a mean half-life of 5.8 hours, hence
the use of a 6-hour dosing interval.187 Indomethacin, largely eliminated through hepatic metabolism in nonpregnant
women, is eliminated unmetabolized during pregnancy.188 The drug is 90% bound to albumin and binds extensively to tissue.189 Maternal, Fetal, and Neonatal Effects. In clinical trials of indomethacin for preterm labor, maternal side effects
have been rare. Reported side effects of indomethacin include platelet
function abnormalities, producing a prolonged bleeding time and
an increased risk of postpartum hemorrhage; acute transient renal insufficiency190; gastrointestinal nausea, vomiting, and diarrhea; and headaches and dizziness.191 Patients with a history of aspirin allergy should not receive other nonsteroidal
anti-inflammatory drugs because they have a high cross-reactivity
with aspirin allergy. Other medical conditions that contraindicate
their use include gastrointestinal ulcerative disease, hepatic or
renal dysfunction, and coagulopathies. Indomethacin is known to cross the placenta.192 Of greatest concern, explaining the lack of widespread acceptance of indomethacin
as a tocolytic agent, are the reported effects of indomethacin
on the fetus and neonate. Prostaglandin synthetase inhibitors, especially
aspirin, have been reported to cause premature closure of the
ductus arteriosus in the fetus and have been associated with pulmonary
hypertension as well as persistent fetal circulation in the neonate.193, 194, 195 Doppler ultrasound studies of the fetal ductus arteriosus during indomethacin
treatment of the mother have revealed transient ductal constriction.196, 197 The frequency of ductal constriction is greater with increasing gestational
age, occurring in 60% of fetuses between 31 and 34 weeks' gestation, in 40% of
fetuses between 27 and 30 weeks' gestation, and in 0% of
fetuses less than 27 weeks' gestation.198 Ductal constriction also is associated more with long-term use of prostaglandin
synthetase inhibitors than with short-term use.199, 200 Recently an increased incidence of other neonatal complications that could
be related to antenatal use of indomethacin has been reported. These
complications include bronchopulmonary dysplasia,201 necrotizing enterocolitis, grades II to IV intracranial hemorrhage, and
persistent ductus arteriosus.202 Because none of these complications have been noted in any of the controlled
clinical trials, and because these complications also are seen
secondary to prematurity, corroboration from other studies is necessary. Also observed with use of prostaglandin synthetase inhibitors is oligohydramnios
caused by a decrease in fetal urinary output occurring within 5 hours
of indomethacin treatment.203, 204 This effect of indomethacin has been associated with perinatal death and
neonatal anuria.205, 206 Usage Guidelines. Used as a tocolytic, an initial loading dose of 50 to 100 mg indomethacin
can be administered either orally or as a rectal suppository, followed
by 25 mg given orally every 6 hours. Concern over the effects of
indomethacin on the fetus correlated to gestational age and prolonged
use has prompted most clinicians to limit use of indomethacin to pregnancies
less than 32 weeks' gestation and for periods of 24 to 48 hours, although
repeated courses or longer courses of therapy have been reported. Ultrasound
assessment of amniotic fluid volume and Doppler assessment
of the ductus arteriosus is recommended after 48 to 72 hours of
use. Clinical Efficacy. The efficacy of indomethacin as a tocolytic agent was first reported in
an uncontrolled observational study.207 This study showed that indomethacin arrested uterine contractions in 80% of
patients within 2 hours of treatment. As with other tocolytic agents, therapy
was more successful if the cervix was dilated less than 3 cm, delivery
being postponed until a birth weight of greater than 2500 g
was achieved in 89% of these patients. In a large observation study, Dudley
and associates208 reported on a series of 167 patients treated with indomethacin for preterm
labor. Of these 167 patients, 79% had delivery postponed for more
than 72 hours, and 67% had delivery postponed for more than 1 week. In
the infants delivered after therapy, there were no cases of premature
closure of the ductus, persistent fetal circulation, or neonatal bleeding
disorders. Niebyl and co-workers conducted a prospective randomized placebo-controlled
study of oral indomethacin administered for a 24 hour period as therapy
for preterm labor.209 Indomethacin was found to be considerably better than placebo and delayed
delivery for 24 hours in 80% of patients. No evidence of premature
closure of the ductus or pulmonary hypertension was found in the infants
of the indomethacin-treated group. In a second randomized clinical
trial of indomethacin compared with placebo, a 7-day delay of delivery
was seen in 83% of indomethacin-treated patients compared with 17% of
placebo-treated patients.210 Trials comparing indomethacin with ritodrine have shown equal efficacy, but
fewer maternal side effects in the indomethacin-treated groups.200, 211, 212 These trials also first reported serious neonatal side effects, including
pulmonary hypertension, bronchopulmonary dysplasia, and necrotizing
enterocolitis occurring in controlled clinical trials. Indomethacin
also has been compared with magnesium sulfate and has exhibited equal
clinical efficacy, but fewer maternal side effects.213 Currently the place of indomethacin in the obstetrician's armamentarium
of tocolytic drugs remains controversial. However, in carefully
selected patients in whom standard tocolytic agents have not succeeded
in producing uterine relaxation and fetal maturity has not been achieved, especially
if the fetal gestational age is less than 28 weeks or
an immature amniotic fluid lecithin/sphingomyelin ratio has been shown, use
of short-term indomethacin is more than justified.214 Indomethacin is possibly also the drug of choice for patients whose pregnancy
is complicated by polyhydramnios and preterm labor.215 CALCIUM-CHANNEL BLOCKERS. Pharmacology. Primarily affecting the voltage-dependent calcium channels within the
cell membrane and inhibiting the influx of Ca2+ into the cell, the slow-channel calcium blockers or calcium antagonists
inhibit smooth muscle contraction.216 Within the cell, the calcium-channel blockers also inhibit release of
Ca2+ from the sarcoplasmic reticulum and promote Ca2+ efflux from the cell.217 These slow-channel calcium blockers are used primarily in the treatment
of cardiac arrhythmias, ischemic heart disease, and hypertension. Verapamil
was first examined as a tocolytic agent and was shown to impair
atrioventricular conduction before effecting uterine tocolysis.218 Nifedipine, however, has proved effective as a tocolytic agent in small
clinical trials.219, 220, 221 Nifedipine is absorbed rapidly after oral administration.222 Sublingual absorption is incomplete and actually is caused by swallowing
and gastrointestinal absorption. In the preterm patient, peak serum
concentrations after sublingual administration show wide variation and
a mean half-life of 81 minutes.223 Placental transfer occurs with a fetalto-maternal concentration ratio
of 0.93 ± 0.2.224 Maternal, Fetal, and Neonatal Effects. Maternal treatment with nifedipine causes peripheral vasodilation with
small decreases in diastolic blood pressure and hemodilution.224 Symptoms reported with use include nausea, flushing, headache, and dizziness. A
rare case of hepatotoxicity has been reported with maternal
use for premature labor.225 Concomitant use of nifedipine and magnesium sulfate has produced potentiation
of hypotension and neuromuscular blockade.226, 227 Nifedipine has been shown to cause a decrease in uterine blood flow accompanied
by in utero fetal hypoxemia, fetal acidosis, and fetal death
in sheep, goats, and monkeys given calcium-channel blockers.228, 229, 230, 231 However, in the small clinical trials using nifedipine for preterm labor, no
adverse fetal or neonatal effects have been reported.232 Usage Guidelines. Nifedipine has been administered orally, 30 mg being given initially, followed
by 20 mg every 6 hours. Sublingual doses of 10 mg every 20 minutes
to a maximum first-hour dose of 40 mg also have been administered. There
have been no studies comparing dosing regimens. Clinical Efficacy. Randomized clinical trials of nifedipine have used the β-mimetics
as comparative tocolytic agents,232, 233, 234 although one study did include an untreated group.221 Success of therapy for all these clinical trials was defined as delay
of delivery for at least 48 hours. In the trial reported by Read and Wellby,221 nifedipine had a higher success rate than either ritodrine or placebo
in the treatment of preterm labor (75% vs 45% and 29%, respectively). Their
trial has been criticized, however, because of methodologic errors. Three
other prospective randomized studies have all found that nifedipine
is as efficacious as ritodrine, but has fewer maternal side effects.232, 233, 234 Because of the relatively few clinical trials published on nifedipine
use in preterm labor, its clinical usefulness is uncertain.45 OXYTOCIN RECEPTOR ANTAGONISTS. Pharmacology. Many small peptide analogs have been developed based on the known structure
of oxytocin and have been modified at various amino acid positions
to change the peptide's activity. These peptides, called oxytocin
receptor antagonists, have been shown to inhibit uterine contractions
in vitro in myometrium obtained from both animals235, 236 and humans237 and in vivo in rats, guinea pigs, and baboons.235, 238 These antagonists act by competing with oxytocin at its myometrial and
decidual receptors, inhibiting release of the second messenger and thereby
blocking an increase in the intracellular free calcium and thus
uterine contractions.239 The peptide analog that has undergone the most development, termed atosiban, differs from oxytocin in the 1, 2, 4, and 8 amino acid positions and
possesses uterine tocolytic activity.240, 241 Maternal, Fetal, and Neonatal Effects. Oxytocin receptor antagonists are theoretically organ specific, which
may minimize their side effects. Placental transfer of atosiban has been
studied in humans undergoing cesarean delivery at term and was found
to be minimal.242 Atosiban had no demonstrable effects on umbilical cord blood pH. Clinical Efficacy. There are two observation studies of atosiban in the literature. In the
first study, atosiban was administered by intravenous infusion in doses
ranging from 10 to 100 μg/minute for 1 to 10 hours and produced
a decrease in uterine activity in all patients.243 In the second study, an additional 12 patients were treated, yielding
a 75% inhibition of uterine contractions and cervical progression.244 Three patients, all at 27 weeks' gestation, failed to respond to therapy, which
the authors believed to be possibly due to decreased myometrial
oxytocin receptor concentration. One randomized clinical trial has
been published.245 In this study the effect of atosiban was measured by the frequency of
uterine contractions. Atosiban produced a 55% mean decrease in contraction
frequency compared with a 27% mean decrease in contraction frequency
seen with the placebo. In this study, tocolytic response did not correspond
with gestational age. Atosiban remains an investigational tocolytic
agent. |