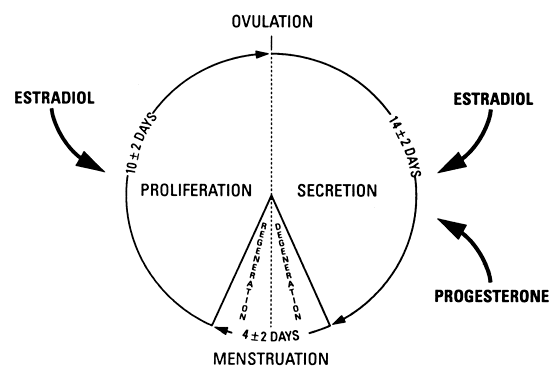
The major morphologic criteria useful for dating the endometrium throughout
the cycle are presented in Figure 3. In routine dating, the pathologist should avoid bias by evaluating the
histologic section before reading the clinical information. After examining
the specimen, the pathologist should attempt to correlate histology
with clinical history. Given histologic expertise, however, the
endometrial biopsy is sufficiently accurate and objective that the date
of the sample seldom has to be changed in the face of a contrary history. In
the most common terminology for dating the endometrial biopsy, day 1 is
used as the first day of bleeding, and this is used in Figure 3 as the starting point.5 It is not necessary to date the endometrium during the proliferative phase. During
this period, daily morphologic alterations are not sufficiently
sensitive. Also, because proliferation precedes the ovulatory period, dating
proliferative endometrium gives the clinician no relevant
information on whether ovulation is occurring. The daily changes in
the endometrium during the postovulatory period, however, are significant
enough from one day to another to provide accurate evaluation of the
endometrial cycle. 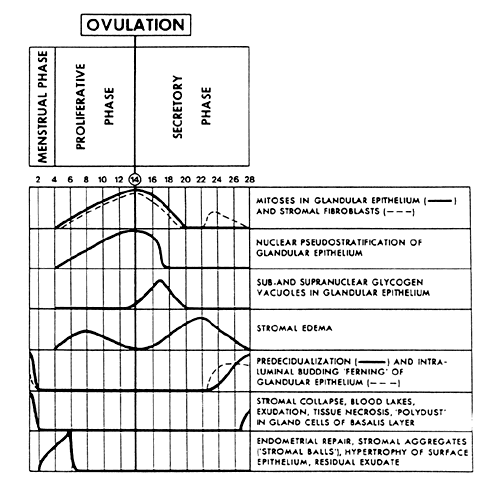 Fig. 3. Major morphologic criteria for dating the endometrium during the menstrual
cycle.(Noyes RW, Hertig AT, Rock J: Dating the endometrial biopsy. Fertil Steril 1:3, 1950. Reproduced
with permission of the publisher. The American
Fertility Society) Fig. 3. Major morphologic criteria for dating the endometrium during the menstrual
cycle.(Noyes RW, Hertig AT, Rock J: Dating the endometrial biopsy. Fertil Steril 1:3, 1950. Reproduced
with permission of the publisher. The American
Fertility Society)
|
Menstrual Period In most women with normal-length cycles, the menstrual period lasts 4 days ± 1 day. During
this period, the endometrial mucosa undergoes
rapid degeneration and regeneration. Both phenomena are presumably
independent of hormonal influence.6 On cycle days 28 and 1, the endometrium is thick, red, and soft. The stroma
beneath the surface epithelium contains blood lakes, fragmented stromal
cells, and inflammatory exudate (Fig. 4A). These rapidly become generalized, resulting in a rough, friable, hemorrhagic
surface. When seen in cross-section, the functional layer of
the endometrium occupies the upper two thirds of the entire thickness; the
lower third, or basal layer, changes very little during the endometrial
cycle. 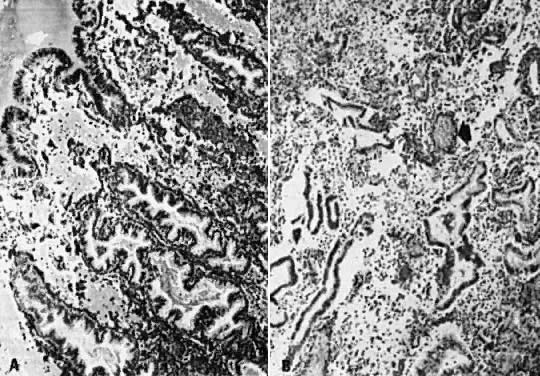 Fig. 4. Menstrual endometrium. A. Menstrual endometrium, day 28 to day 1, with collapsed stroma and blood
lakes beneath the surface epithelium ( arrow) (× 100). B. Menstrual endometrium, day 1 to day 2, showing ruptured glands with secretory
exhaustion, degenerated predecidua, inflammatory cells, and thrombosed
vessels ( arrow) (×100). Fig. 4. Menstrual endometrium. A. Menstrual endometrium, day 28 to day 1, with collapsed stroma and blood
lakes beneath the surface epithelium ( arrow) (× 100). B. Menstrual endometrium, day 1 to day 2, showing ruptured glands with secretory
exhaustion, degenerated predecidua, inflammatory cells, and thrombosed
vessels ( arrow) (×100).
|
On cycle day 2, the functional layer becomes disorganized, containing predecidual
stromal cells admixed with epithelial glandular cells; both
cellular systems undergo severe degenerative changes (Fig. 4B). These are diffuse throughout the endometrial mucosa of the body and
fundus regions. The isthmic endometrium is not significantly sensitive
to cyclic hormonal stimuli and is not used for dating purposes. The menstrual
fluid is made up of autolyzed tissue admixed with a heavy polymorphonuclear
exudate, red blood cells, and proteolytic enzymes. One
of the latter is blood protease plasmin, which prevents clotting of menstrual
blood. Plasminogen activators, which convert plasminogen into
plasmin, are found in and released from degenerated endometrial vascular
endothelium.7 Anovulatory bleeding, in contrast to menstrual bleeding, is associated
with focal tissue breakdown and thrombosis of dilated vessels. The adjacent
endometrium is intact and most commonly is of the hyperplastic
type. During cycle days 1 and 2, synthesis of nuclear deoxyribonucleic acid (DNA) is
near zero level in the secretory functional layer of the endometrium. These
findings are consistent with previous ultrastructural observations, indicating
that the cellular components of the functional
layer undergo irreversible cell injury before being expelled during the
menstrual period.6 On cycle days 2 and 3, the functional layer gradually becomes cleaved off
from the underlying basal layer, resulting in a thin, denuded basal
layer with a ragged surface onto which residual basal gland stumps open (Fig. 5A). Loss of the bulk of endometrial mucosa explains why scant tissue is
obtained in endometrial biopsy or dilation and curettage (D & C) during
the second part of the menstrual period. 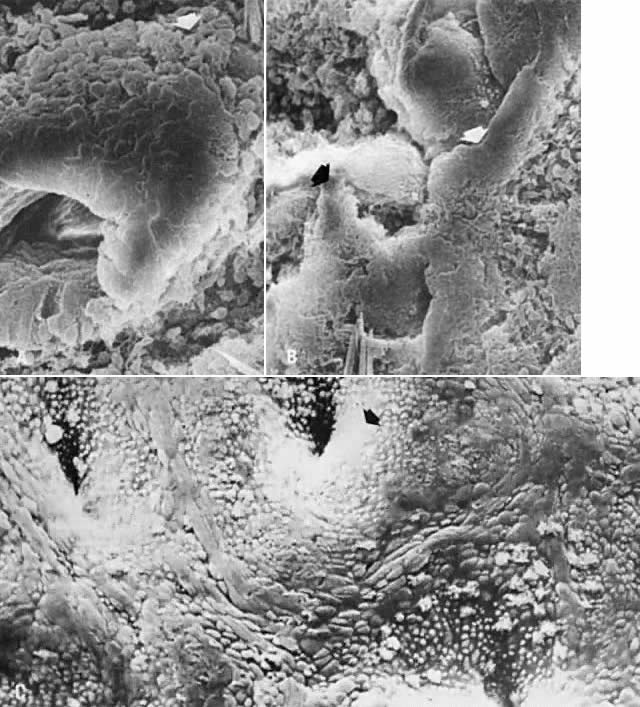 Fig. 5. Scanning electron microscopy of regenerative endometrium. A. On cycle day 3, gland stumps of residual basal glands have epithelial
extensions ( arrow) onto denuded stroma (×380). B. Cycle day 4 is dominated by anastomoses of epithelial membranes ( arrows) from adjacent basal gland openings (×230). C. By cycle day 5, the surface is fully regenerated. Note concentration of
ciliated cells ( arrow) around gland openings (×300). Fig. 5. Scanning electron microscopy of regenerative endometrium. A. On cycle day 3, gland stumps of residual basal glands have epithelial
extensions ( arrow) onto denuded stroma (×380). B. Cycle day 4 is dominated by anastomoses of epithelial membranes ( arrows) from adjacent basal gland openings (×230). C. By cycle day 5, the surface is fully regenerated. Note concentration of
ciliated cells ( arrow) around gland openings (×300).
|
Starting on day 2 and for the subsequent 2 days, proliferation of the basal
gland epithelium begins in the areas of denudation.6 The surface of the endometrium is re-epithelialized as the residual glandular
epithelium spreads over the denuded surface (see Fig. 5A). Another source of resurfacing epithelium is the surface epithelium of
peripheral regions of the endometrial cavity, such as the lower uterine
segment and peritubal ostia, which remain intact during the menstrual
period.6 The subsequent development of interanastomoses between converging epithelial
proliferations (Fig. 5B) leads ultimately to complete reconstruction of a new surface epithelium
by cycle day 5 (Fig. 5C). Complete re-epithelialization of the surface coincides with cessation
of bleeding. DNA tracing studies have shown that increased DNA synthesis is confined
to the basal layer of the body and fundus of the uterus and the adjacent
isthmic and peritubal-ostial endometrial mucosa, all of which remain
intact during menstruation.6 This increase occurs only after complete denudation of the basal layer
by cycle day 3. The postmenstrual endometrium is repaired by cellular migration and replication
of surface epithelial cells.6 The resurfacing cells are flattened and spindle-shaped, with intracellular
microfilamentous-microtubular systems and pseudopodial projections (Fig. 6). These features are consistent with ameboid contraction-expansion-mediated
motility. 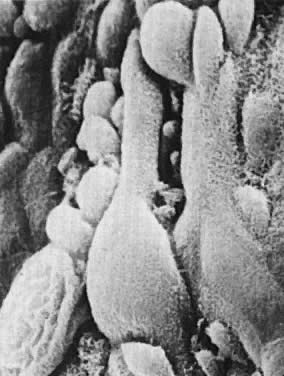 Fig. 6. Regenerative endometrium. The newly formed epithelial cells have flattened
cytoplasm and prominent pseudopodial projections (×24,000). Fig. 6. Regenerative endometrium. The newly formed epithelial cells have flattened
cytoplasm and prominent pseudopodial projections (×24,000).
|
Endometrial stromal cells modulate the growth, steroid hormone action, and
functional differentiation of the epithelial cells.3 In vitro experiments have demonstrated production of basement membrane-like
material with heavy desmosomal attachments by epithelial (but not
stromal) cells. The basement membrane provides anchorage and participates
in the control of proliferation and migration of epithelial cells
as well as in their differentiations.8 Tenascin, an extracellular matrix protein, is immunolocalized around proliferative
endometrial glands and is believed to enhance epithelial
cell migration and proliferation during periods of postmenstrual repair
by inhibiting cell attachments to fibronectin.9 After the initial epithelial spread, cell division and migration operate
simultaneously until a confluent surface layer has been regenerated
by cycle day 5. The sudden increase in nucleic acid synthesis and a very
short DNA-synthesis phase of the regenerative cells result in accelerated
tissue turnover (Fig. 7). These characteristics of migration and accelerated tissue turnover explain
the spectacularly rapid wound healing capability of the human endometrium. DNA
and ultrastructural data do not support the concept that
the regenerative endometrium derives directly from persistent secretory
spongiosa or stromal fibroblasts of the endometrium. 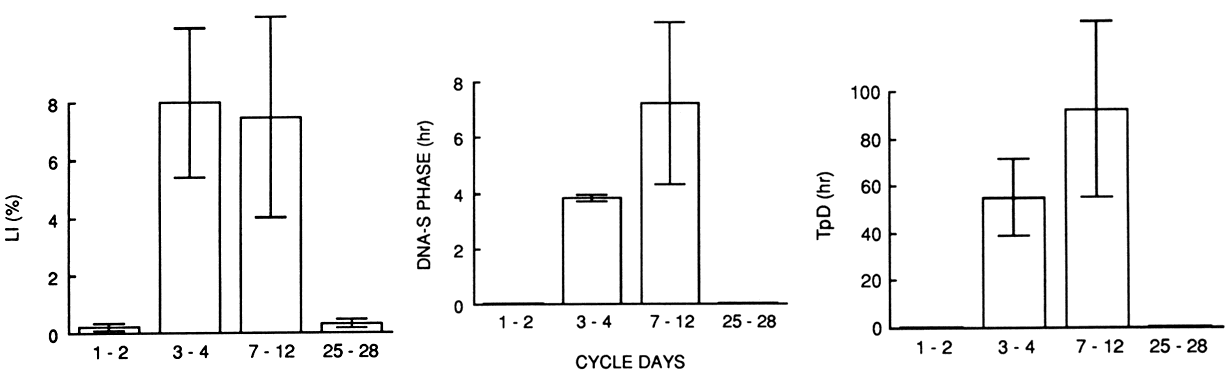 Fig. 7. Kinetic characteristics of the endometirum during the menstrual cycle according
to in vitro historadioautography using the double-labeling technique
with 3 H-thymidine. Labeling index ( LI ), DNA synthesis phase ( DNA-S phase ), and potential doubling time ( TpD) are negligible during the premenstrual and early menstrual periods. Note
the sudden increase in LI and shortening of the DNA-S phase and tissue turnover time during the regenerative period on cycle days 3 to 4. The
postregenerative period (cycle day 5 on) is characterized
by prolongation of both the DNA-S phase and tissue turnover time.(Ferenczy A: How to date the endometrial cycle. Contemp Obstet Gynecol 18:115, 1981) Fig. 7. Kinetic characteristics of the endometirum during the menstrual cycle according
to in vitro historadioautography using the double-labeling technique
with 3 H-thymidine. Labeling index ( LI ), DNA synthesis phase ( DNA-S phase ), and potential doubling time ( TpD) are negligible during the premenstrual and early menstrual periods. Note
the sudden increase in LI and shortening of the DNA-S phase and tissue turnover time during the regenerative period on cycle days 3 to 4. The
postregenerative period (cycle day 5 on) is characterized
by prolongation of both the DNA-S phase and tissue turnover time.(Ferenczy A: How to date the endometrial cycle. Contemp Obstet Gynecol 18:115, 1981)
|
The mechanisms of induction of endometrial proliferation during menstruation
do not seem to be influenced by estradiol. Indeed, during cycle
days 3 and 4, despite increased DNA activity, plasma levels of estrogens
and progesterone receptors are low and unchanged from the premenstrual
values (see Fig. 7). Also, in experimental endometrial regeneration in the rabbit, proliferation
kinetics and morphologic alterations of the regenerative but estrogen-deprived
atrophic endometrium associated with ovariectomy are
similar to those in animals with intact ovaries.6 In contrast, cycle days 7 through 12 are characterized by a sudden increase
in the incorporation of nuclear thymidine (see Fig. 7; Fig. 8) and mitoses in the stromal, vascular, and gland cell components of the
regenerated endometrium. These changes are accompanied by an increase
in plasma levels of estrogens and progesterone receptors and a slight
decrease in serum pituitary hormones (see Fig. 7), alterations that are consistent with target cell sensitivity and response
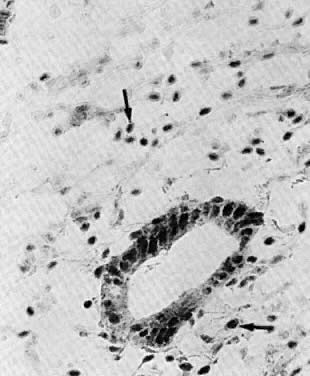
to preovulatory estradiol. 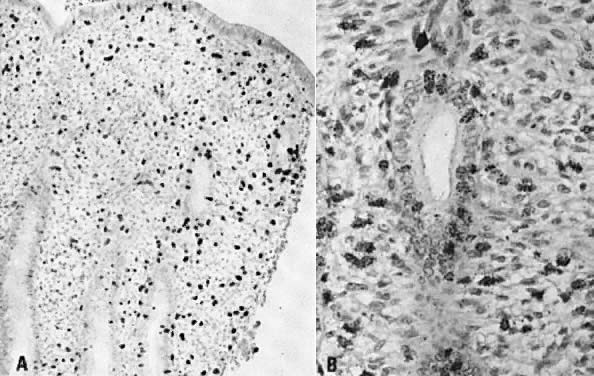 Fig. 8. Historadioautography of proliferative endometrium. A. Note heavy labeling of functional layer of endometrium (×100). B. Detailed view of radiothymidine granules in nuclei of endometrial gland
cells, stromal fibroblasts, and capillary endothelium ( arrow) (×250). Fig. 8. Historadioautography of proliferative endometrium. A. Note heavy labeling of functional layer of endometrium (×100). B. Detailed view of radiothymidine granules in nuclei of endometrial gland
cells, stromal fibroblasts, and capillary endothelium ( arrow) (×250).
|
Proliferative Phase The preovulatory endometrium demonstrates proliferative changes in the
glands, stromal cells, and vascular system.1,4,5,10 The glands acquire numerous mitoses and become longer and larger, with
convoluted shapes. The stroma becomes vascularized. These changes take
place under the stimulatory action of estradiol, which stimulates the
DNA-promoter enzyme, thymidylate synthetase.7,10,11 This, in turn, stimulates ribonucleic acid (RNA) polymerase activity,12 DNA synthesis, and mitosis. Increased proliferation leads to a considerable
thickening of the endometrial mucosa. It is interesting to note
that the endometrium demonstrates geographic variations in its response
to hormonal stimuli. Maximum DNA synthesis is observed in the fundus
and body of the uterus, whereas the isthmic and cornual regions contain
comparatively lower values.10 Also, nuclear DNA activity is higher in the upper third than in the lower
two thirds of the functional layer.10 This zonal variation in sensitivity of endometrial tissue to hormonal
influence may be related to different physiologic functions: the upper
functional layer facilitates implantation and nutrition of the blastocyst, whereas
the lower functional layer is involved in the secretory
activity and provides for the integrity of the endometrial mucosa. Whether
differences in hormonal responses are due to dissimilar vascular
supply of the upper and lower layers or to the intrinsic, heterogeneous
nature of the endometrial tissue in terms of receptor content or to
both remains to be determined. Maximum DNA synthesis during the midproliferative
phase of the cycle (i.e., cycle days 8 to 10),10 correlates with the maximum number of mitoses in both the stromal and
epithelial endometrial cellular elements as well as with the midproliferative
peak of plasma estradiol and nuclear estradiol receptors (see Fig. 7).13 Increased nuclear DNA synthesis and mitotic activity in gland cells correlate
with high levels of nucleolar organizer regions. These are loops
of DNA, transcribed to ribosomal RNA, and are proliferation indicators.9 An
increase in estradiol-mediated DNA activity during the early and
midproliferative phases and its decrease by cycle days 11 through 14 may
reflect the growth-inhibition effects of long-standing estradiol
stimulation. According to animal studies,14 DNA synthesis decreases rather than increases after 2 days of estrogen
administration. Inhibition of nucleic acid synthesis is apparently not
related to loss of estradiol receptors or nuclear translocation of estradiol
receptors, but rather, presumably, to accumulation of the chalone-like
inhibitors of DNA synthesis.14 This hypothesis is attractive, but it remains to be tested. Recent biochemical and immunohistochemical studies have identified several
peptides suspected to be involved in the autocrine or paracrine control
of endometrial growth.15 Epidermal growth factor is produced by both epithelial and stromal cells
and is likely to stimulate alone or with estradiol stromal/epithelial
proliferation. Insulin-like growth factors also promote endometrial
cellular mitosis, including that of decidual cells. It is likely that
epidermal growth factor mediates the mitogenic action of estradiol. The
transforming growth factor-β mainly inhibits cellular proliferation
and promotes differentiation of the endometrium, acting via autocrine
and paracrine mechanisms. It is increased in the secretory phase
and particularly in early pregnancy decidua and may enhance placental
calcium transport. Cytokines are multifactorial immunomodulators acting
by autocrine, paracrine, or endocrine mechanisms on proliferation and
differentiation of cells of the immune system. In addition to tissue proliferation, estradiol promotes the development
of free and bound ribosomes, mitochondria, golgiosomes and primary lysosomes
in gland cells and presumably in stromal cells (Fig. 9A). Biochemically, these organelles each provide for protein matrix, energy, and
synthesis of various enzymes. Some of these enzymes, including
glucose-6-phosphatase, hexokinase, pyruvate kinase, and lactate dehydrogenase, are
involved in carbohydrate metabolism.16 Other important proteins thought to be produced by estradiol are estradiol
receptors and progesterone receptors.11,17 Concentrations of estradiol receptors and progesterone receptors increase
in both the blood and the endometrium during the proliferative phase
of the cycle (see Fig. 7). Another characteristic feature of proliferative-surface and gland-lining
cells is an increase in the number of cilia and microvilli (Fig. 9B-C). These decrease considerably during the secretory phase, suggesting
that endometrial ciliogenesis and microvillogenesis are estrogen dependent.16 Additional evidence for this concept is provided by observations of even
more cilia and microvilli in hyperplastic endometria, whereas progestational
therapy leads to their disappearance and decrease, respectively.16 Ciliated cells are especially numerous around gland openings (see Fig. 5C). It has been suggested that this peculiar distribution and strong-forward
and slow-recovery ciliary beat pattern facilitate mobilization and
distribution of endometrial secretions during the luteal phase of the
cycle.16 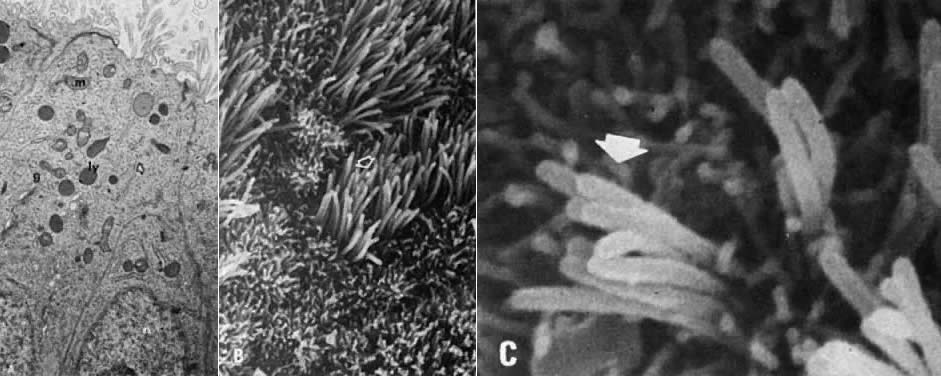 Fig. 9. Proliferative endometrium. A. Transmission electron microscopy of gland cells with numerous supranuclear (n), golgiosomes (g), mitochondria (m), granular endoplasmic reticulum ( arrow ), and electron-dense primary lysosomes ( ly) (×13,000). B. Surface epithelium with numerous ciliated ( arrow) and nonciliated microvillous cells (×3000). C. High magnification of ciliary shafts ( arrow) in the process of development (×10,000). Fig. 9. Proliferative endometrium. A. Transmission electron microscopy of gland cells with numerous supranuclear (n), golgiosomes (g), mitochondria (m), granular endoplasmic reticulum ( arrow ), and electron-dense primary lysosomes ( ly) (×13,000). B. Surface epithelium with numerous ciliated ( arrow) and nonciliated microvillous cells (×3000). C. High magnification of ciliary shafts ( arrow) in the process of development (×10,000).
|
Intracytoplasmic filaments serve as a cytoplasmic “skeleton” in
gland and stromal cells. Gland cells have cytokeratin- and vimentin-positive
intracytoplasmic filaments, whereas endometrial stromal cells
contain vimentin and smooth muscle-related antigens.4 Lymphoid aggregates resembling follicles may be seen in the endometrial
stroma, particularly in the basal layer and during the proliferative
phase of the cycle.18 They are made of T-cells, macrophages, and B-cells16 and stain for IgA, IgM, or IgG. They are unlikely to play a significant
role, if any, in the local secretory immune system. Indeed, endometrial
epithelial cells synthesize negligible amounts of immunoproteins,19 and IgG-containing plasma cells are absent in normal endometrium. The
observations are consistent with the sterile nature of normal endometrium. Secretory Phase During the postovulatory phase, or secretory phase, the estradiol-primed
endometrium is under progestagenic stimulation and undergoes secretory
differentiation.4,5,16 The first morphologic event indicating ovulation occurs on the 16th day
of the cycle (postovulation day 2, or POD 2) with the appearance of
small, cylindric vacuoles occupying the base of some of the gland cells
in the functional layer. Because similar changes may be produced by
estrogens alone in the absence of ovulation, incomplete or abortive subnuclear
vacuolization is not considered specific to ovulation. The first
reliable histologic alterations that are considered specific to ovulation
are seen on the 17th day (POD 3) of the cycle.4,5,16 These include well-developed subnuclear glycogen vacuoles in gland lining
cells and palisading of gland cell nuclei. Both phenomena involve
every cell in a given gland (Fig. 10A). TAG-72 or B72.3, a mucin-like glycoprotein, is exclusively found in
postovulatory secretory gland cells; it serves as an immunomarker of ovulation.4 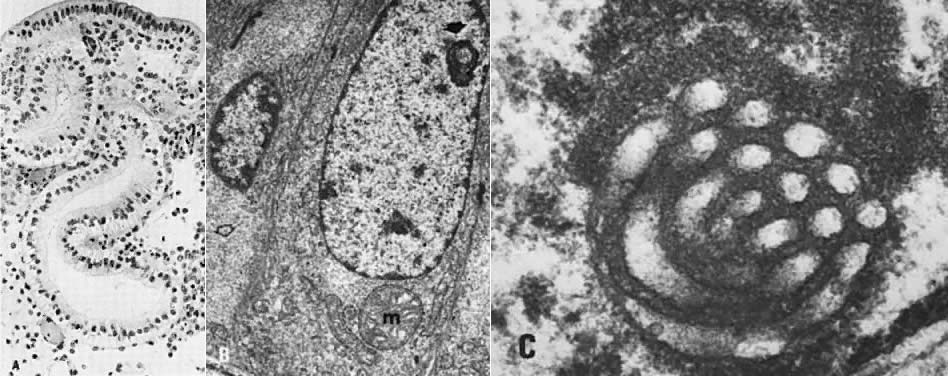 Fig. 10. Secretory endometrium. A. Day 17 (POD 3) secretory endometrial glands with S-shaped configuration, subnuclear
vacuolization, and palisading of nuclei in middle of lining
epithelium (×450). B. Ultrastructurally, ovulation is followed by appearance of giant mitochondria (m), nucleolar
channel system ( arrow ), and abundant glycogen granules ( open arrow) in gland cells (×7100). C. Detailed view of nucleolar channel system made of hollow tubules and embedded
into nucleolar ribosomes (×30,000). Fig. 10. Secretory endometrium. A. Day 17 (POD 3) secretory endometrial glands with S-shaped configuration, subnuclear
vacuolization, and palisading of nuclei in middle of lining
epithelium (×450). B. Ultrastructurally, ovulation is followed by appearance of giant mitochondria (m), nucleolar
channel system ( arrow ), and abundant glycogen granules ( open arrow) in gland cells (×7100). C. Detailed view of nucleolar channel system made of hollow tubules and embedded
into nucleolar ribosomes (×30,000).
|
At the transmission electron microscopic level, ovulation may be recognized
by the appearance of giant mitochondria and the so-called nucleolar
channel system in gland cells.16 Nucleolar channel systems are unique to women and occur only during the
postovulatory period (Table 1; Fig. 10B-C). They are presumably produced by the infolding of the nuclear membranes
under progesterone stimulation. TABLE 1. Morphologic Evidence of Ovulation
| Cycle |
Morphology | Days |
Nucleolar channel system* in gland cells | 15–25 |
Subnuclear vacuolization with nuclear palisading of gland cells | 17–18 |
Stromal edema with ferning of glandular epithelium | 22–23 |
Perivascular and stromal predecidulalization | 23–28 |
Diffuse predecidual and glandular necrosis, inflammation, and vascular
thrombosis | 1–2 |
Inflammatory exudate, aggregates of stromal cells (stromal balls) with
or without | 2–4 |
hypertrophic surface epithelial cells, diffuse | |
* At ultrastructural level.
During the first 4 postovulatory days, occasional mitoses are seen in the
glandular epithelium. The glands are engaged in intracellular synthesis, but
not as yet in active extracellular secretion of glycoproteins. On
cycle days 19 and 20 (PODs 5 and 6), the intracellular secretory
products are extruded into the glandular space by apocrine-type secretion. This
is characterized by protrusions and eventual detachment of
the apical portion of cells containing glycoproteins. Transudation of
plasma from circulating blood in the endometrial mucosa also contributes
to uterine secretory fluids. The peak of intraglandular secretions
on cycle day 21 (POD 7) coincides with the time of implantation of the
free blastocyst if fertilization has taken place in this cycle. Nucleic
acid synthesis by gland cells ceases as apocrine secretory activity
is initiated by day 19 (POD 5) (see Fig. 7). This correlates with total lack of mitoses in the glands during the
mid and late periods of the secretory phase.4,5,16 Inhibition of mitosis has been attributed to rising levels of postovulatory
progesterone, which antagonizes the action of estradiol by decreasing
estrogen receptors17 and by increasing the progesterone-specific enzyme 17β-hydroxydehydrogenase.13 This enzyme converts estradiol into estrone, which leaves the target cell
with negligible stimulatory effect on the nucleus.13 As a result, an increase in 17β-hydroxydehydrogenase lowers the concentration
of estradiol and its products (i.e., specific receptors for estradiol) in the tissue and increases estrone
sulfotransferase.1,2 It has been suggested that in mice, progesterone induces the epithelial
cells to enter a nondividing (G0) stage of the cell cycle during decidualization.20 For accurate dating purposes from cycle day 21 (POD 7) on, the pathologist
relies on changes in the stroma, rather than in the glands. These
include edema, coiling of spiral arterioles, and predecidualization of
the stroma (Fig. 11A-C). These alterations are possibly mediated by prostaglandin F2α (PGF2α ) and prostaglandin (PGE2). Indeed, estradiol stimulates the production of PGF2α, whereas progesterone stimulates the synthesis of both PGF2α and PGE2 in vitro.21 PGE2 presumably promotes capillary permeability and stromal edema via histamine, whereas
vascular endothelial growth factor is a potent mitogen of
endothelial cells. Endometrial vascular proliferation at the implantation
site is related to the blastocyst rather than to histamine or PGE2. The blastocyst has a unique biologic property that is shared only with
tumor cells producing the so-called angiogenesis factor, a substance capable of inducing growth of new capillaries.22 Receptors for human chorionic gonadotropin and luteinizing hormone are
present in vascular smooth muscle cells and endothelium, suggesting their
possible role in regulating blood flow.23 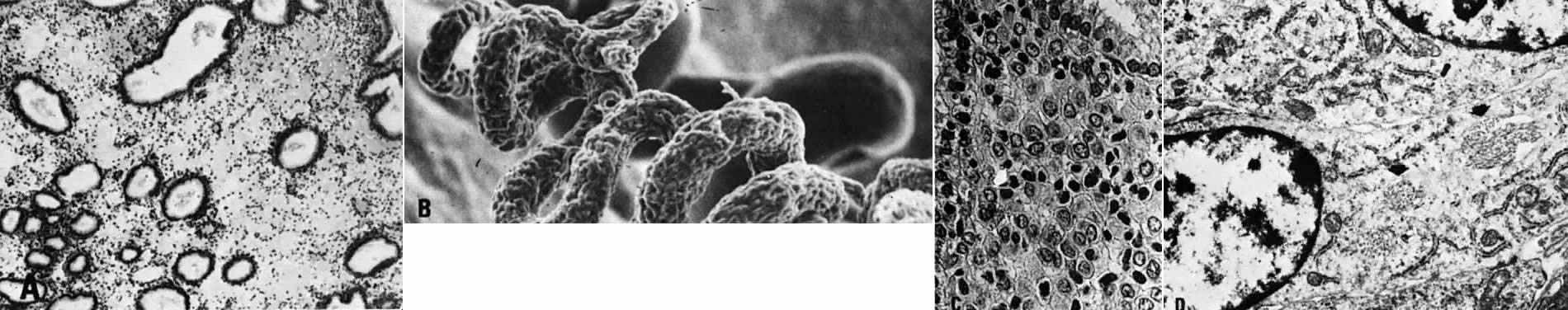 Fig. 11. Secretory endometrium. A. Cycle day 22 (POD 8) characterized by marked stromal edema (×50). B. Scanning electron microscopy of spiral arteriole on cycle day 23 (×100). C. Predecidual cells with large, round nuclei and cytoplasm. Note well-defined
cellular membranes producing a “honeycomb” pattern. Mitoses
are seen ( arrow) (×450). D. Ultrastructure of predecidual cells containing membrane-bound autophagosomes ( arrows) with incorporated collagen fibers (×8000). Fig. 11. Secretory endometrium. A. Cycle day 22 (POD 8) characterized by marked stromal edema (×50). B. Scanning electron microscopy of spiral arteriole on cycle day 23 (×100). C. Predecidual cells with large, round nuclei and cytoplasm. Note well-defined
cellular membranes producing a “honeycomb” pattern. Mitoses
are seen ( arrow) (×450). D. Ultrastructure of predecidual cells containing membrane-bound autophagosomes ( arrows) with incorporated collagen fibers (×8000).
|
PGF2α, Ki-67, and other peptides are responsible for the predecidual transformation
and growth of spindle-shaped stromal cells.24 Estradiol is not involved in this process because decidualized cells have
no receptors for estradiol. This change consists of cytonuclear enlargement
with tetraploid nuclei resulting in plump, liver-like epithelioid
cells (Fig. 11C). Predecidualization (not pseudodecidualization) is accompanied by an
increase in nuclear DNA synthesis, mitotic activity,16 and the formation of a pericellular laminin substance.25 The latter is typical of epithelial cells and is believed to be related
to the midsecretory phase peak of estradiol plasma levels. Although
progesterone plasma levels are also elevated during this period of the
cycle, progesterone-dependent 17β-hydroxydehydrogenase appears to
be a specific enzyme only for the endometrial gland cells.13 Consequently, predecidual cells are relatively independent of the growth-inhibitory
effect of progesterone. Predecidual cells are of stromal origin. They have mesenchymal-related (e.g., vimentin, desmin), not epithelial-related (e.g., cytokeratin) antigens.4 They represent precursor forms of gestational decidual cells (decidua
vera). Because they develop after implantation, they are not involved
in the implantation process per se. The cells have several metabolic functions
related either to pregnancy or, if conception has not occurred, to
menstrual breakdown of the endometrium. For example, prolactin is
produced by decidual cells and is related to osmoregulation of amniotic
fluid.15 After implantation, the decidual cells appear to control the invasive
nature of the normal trophoblast,26,27 for a lack of decidual reaction in the endometrium is accompanied by deep
myometrial implantation of the placenta (i.e., placenta accreta, increta, percreta). Decidual tissue during early periods of gestation is rich in lymphocytes, natural
killer cells and macrophages. The latter are observed near
nidation sites, are of the major histocompatibility complex (MHC) class
II type, and have the potential to present fetal antigens to T-lymphocytes. In
addition, they secrete monokines and have immunosuppressive
activity.28 In the nongestational endometrium, predecidual cells are engaged in phagocytosis
and digestion of extracellular collagen matrix13 (Fig. 11D), and these cellular activities may contribute to menstrual breakdown
of the endometrium. Uterine decidual response may be induced experimentally
both in vivo and in vitro in the prepubertal rat maintained on progesterone
by PGF2α.24 Indomethacin, an inhibitor of prostaglandin synthesis, prevents decidualization, providing
supporting evidence for the concept of prostaglandin-mediated
decidual reaction. The presence of progesterone, however, appears
to be a prerequisite for decidualization, for without it, no
such reaction is observed in vitro.24 Predecidual transformation of stromal cells begins around the arterioles
on day 23 (POD 9) and later involves the stroma under the surface epithelium, producing
the compact layer by day 25 (POD 11). By the 26th (POD 12) day of the cycle, the predecidual stromal reaction
under the surface epithelium and around the arterioles fuses together, forming
large sheets of predecidua. One day later, this confluence may
involve the upper two thirds of the functional layer. In addition to
marked predecidualization, the stroma on cycle days 26 (POD 12) and 27 (POD 13) is
associated with a progressively increasing number of extravasated
polymorphonuclear leukocytes (Fig. 12) and the so-called endometrial granulocytes ( Körnchenzellen), or K-cells.16,29 These granulocytes have a granular, eosinophilic, cytoplasmic substance
and resemble eosinophils except for having a single, round (nonlobulated) nucleus. They
are members of the large granular lymphocyte series. They
are particularly numerous during the first trimester of pregnancy
and may play a role in placentation. By day 28 (POD 14), the spiral
arterioles become dilated, and fissures appear in the compact layer (see Fig. 4A). These fissures contain edematous fluid, red blood cells, and acute inflammatory
exudate. 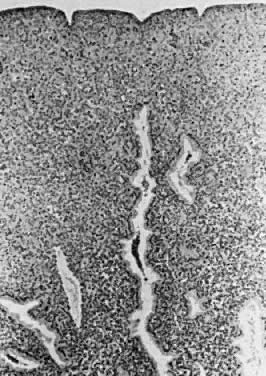 Fig. 12. Secretory endometrium. Cycle days 26 and 27 (PODs 12 and 13) are marked
by predecidualization of the functional layer, which is scattered with
inflammatory cells. Glands demonstrate secretory “exhaustion,” with
inspissated intraglandular secretions (×100). Fig. 12. Secretory endometrium. Cycle days 26 and 27 (PODs 12 and 13) are marked
by predecidualization of the functional layer, which is scattered with
inflammatory cells. Glands demonstrate secretory “exhaustion,” with
inspissated intraglandular secretions (×100).
|
The cyclic endometrium, as with many other tissue systems in the body, is
subject to two fundamental types of cell death: apoptosis and necrosis. Apoptosis, or programmed, multifocal single cell death plays a complementary but
opposite role to mitosis in normal tissue homeostasis. It is considered
to be the major process responsible for cell death in various physiologic
cell-turnover and differentiation events in both the embryo and
the adult.30 Histologically, apoptosis is recognized by multiple fragments of condensed, shrunken
nuclear material and cytoplasm of single cells. These alterations
were traditionally called “polydust” before the
true origin of this nuclear debris was recognized.4 These apoptotic bodies are subsequently engulfed by adjacent cells. An
inflammatory reaction composed of leukocytes is generally absent in association
with apoptotic bodies. Apoptosis is seen in both gland cells
and predecidual cells, both in vivo and in vitro.31 The process is likely to be initiated by increased levels of tumor suppressor
p53 and transforming growth factor-β due to severe (irreversible) DNA
damage. Whether apoptosis is produced by a single mechanism
of endonuclease activation leading to the hallmark of apoptosis, which
is internucleosomal DNA cleavage and subsequently DNA fragmentation
is not clear.32 In vitro experiments in the rodent suggest that apoptosis in endometrial
stromal cells, including their decidual variants, is induced by transforming
growth factor-β2 via an autocrine or paracrine mechanism.31 The extent that apoptosis contributes to endometrial degeneration and
gestational decidual regression is not clear. Also unclear is whether apoptosis is more or less important than tissue necrosis. Indeed, the endometrium demonstrates morphologic evidence of coagulative
tissue necrosis in response to vasoconstriction of basal arteries and
ischemia. The necrotic tissue with increased hydrolytic enzyme activity
is characterized by collapse of the upper two thirds of the functional
layer. It contains tissue fragmentation with swelling of the cells' nuclei
and membrane disruption. The final event is heterolysis of
dead cells due to the action of inflammatory cells invading the necrotic
tissue. Electron microscopy combined with enzyme-tracing studies demonstrates that
in the absence of conception, the endometrium undergoes gradual involution, degeneration, and ischemic necrosis during the last 5 days of
the cycle.5,6,16,33,34 During the proliferative phase, estradiol stimulates the development of
Golgi complex-derived primary lysosomes, many of which contain acid
phosphatase, a potent lytic enzyme (see Fig. 9A). Primary lysosomes are present in the epithelial, stromal, and endothelial
cells of the functional layer of the endometrium.16,33 During the first half of the luteal phase, lytic enzymes, including acid
phosphatase, are confined to membrane-bound lysosomes. Their release is presumably inhibited by the membrane-stabilizer effect
of progesterone. The sudden decrease in estradiol and progesterone levels
causes a failure in the membrane integrity of acid phosphatase-containing
lysosomes. As a result, the lysosomal enzyme is released into
enzyme-free autophagic bodies filled with sequestered intracellular elements. The
destructive action of the acid hydrolases leads to digestion
of the incorporated cytoplasmic elements, producing empty vacuoles (Fig. 13). Thus, portions of the cytoplasmic substance are removed by lysosomal
autodigestion. It also has been suggested that the gradual increase in
lysosome-membrane permeability may result in direct intracellular and
intercellular diffusion of the lytic enzymes, including type II collagenase
found in predecidual cells. Relaxin, which is found in gland and
decidualized cells, stimulates collagenase and plasminogen activators, contributing
to tissue breakdown.15 It destroys the glandular and stromal cells as well as the vascular endothelium. Vascular
luminal surface membrane injury promotes platelet
deposition, release of prostaglandins, vascular thrombosis, and eventually
tissue necrosis.16 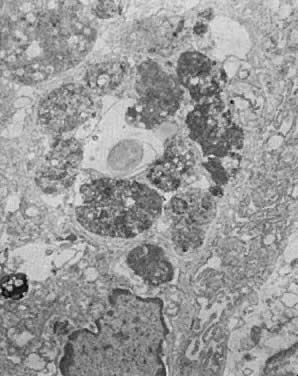 Fig. 13. Secretory endometrium, cycle day 27 (POD 13). Portion of gland cell with
prominent membrane-bound autophagosomes in various stages of enzymatic
digestion of incorporated organelles and membranes. In the middle, an
empty vacuole is seen, indicating total digestion of incorporated material (×9000). Fig. 13. Secretory endometrium, cycle day 27 (POD 13). Portion of gland cell with
prominent membrane-bound autophagosomes in various stages of enzymatic
digestion of incorporated organelles and membranes. In the middle, an
empty vacuole is seen, indicating total digestion of incorporated material (×9000).
|
Recently, a cytoplasmic pore-forming protein, perforin, which is present
in the cytoplasmic granules of cytotoxic T-lymphocytes and natural killer
cells, has been demonstrated in the human endometrium.35 Perforin-positive cytotoxic lymphocytes (CD3, CD56, and CD8) become numerous during the second phase of the postovulatory phase of
the menstrual cycle, and their colonization is progesterone mediated. Perforin
forms pores on the target cell membrane, enhancing passage of
cytotoxic molecules that in turn produce or contribute to target cell
death. Their maximum accumulation before menses and their participation
in the regression of nongestational corpus luteum35 and deciduoma in the rodent may suggest that perforin-positive cytotoxic
cells may play an important role in the menstrual breakdown of the
endometrium. PGF2α and PGE2 increase significantly in the secretory endometrium by the 25th day of
the cycle and reach maximum concentrations during the menstrual period,36 but PGF2α increases to a much greater degree than does PGE2. It has been speculated that the high levels of the potent vasoconstrictors
endothelin, which acts on spiral arterioles, and PGF2α, which is seen during menstruation, may stimulate the onset of bleeding
via vasoconstriction of spiral arterioles at the endometrial-myometrial
junction and the expulsion of degenerated endometrium through myometrial
contractions, respectively.34,37,38 |