Although estrogen is not required for contraceptive efficacy of hormonal methods of contraception, the combination of estrogen and progestin in OCs increased the effectiveness of the method and improved bleeding patterns compared to progestin only OCs. CVR formulations that contain and release both a progestin and estrogen have cyclic patterns of use that mirror combination OCs. The CVRs are worn for 3 weeks and then removed for 1 week to allow withdrawal bleeding. In combination CVRs, the addition of estrogen combined with a higher dose of progestin result in greater effectiveness compared with progestin-only CVRs, similar to combination OCs. In addition, the estrogen component maintains the endometrium and prevents breakthrough bleeding.
Several formulations using different progestins have been investigated for clinical use. Levonorgestrel and estradiol combined in a CVR demonstrated high efficacy, excellent bleeding patterns, and high continuation rates. Clinical studies of two CVRs of different sizes containing levonorgestrel and estradiol confirmed almost complete ovulation suppression with use. The 1-year pregnancy rate was 2.4 per 100 women for a 50-mm CVR and 1.4 per 100 women using a 58 mm CVR.16,17 Continuation rates were also high, with approximately half of CVR users still using the method at 1 year, more than OC users in a randomized trial comparing the two groups.17 There were no clinically significant changes in the lipid panel with this CVR in several studies.18,19,20,21 However, concern regarding the effect on lipids by CVRs containing levonorgestrel and estradiol arose in a study comparing CVRs with five different progestins; progesterone, levonorgestrel, norethisterone, medroxyprogesterone acetate, megestrol acetate.22 All but the progesterone CVR were combined with estradiol. In this trial, use of the CVR containing levonorgestrel and estradiol was associated with a significant decrease in HDL levels and in the LDL-to-HDL ratio, and an increase in LDL levels.22 These changes were not observed with the other CVRs studied.
Animal data on atherosclerotic disease corroborated the association of this CVR with adverse changes in lipids. In a comparative trial, macaques treated with a CVR containing levonorgestrel and estradiol for 2 years had atherosclerotic changes that were greater than those in the controls.23 Therefore, despite the high efficacy, acceptability, and desirable bleeding patterns associated with this CVR, further study of combined hormonal CVRs were directed to other formulations. The lipid effects seen with this combination CVR are presumed to be caused by the use of a 19-nortestosterone derivative progestin (levonorgestrel) combined with a low-potency estrogen (estradiol). Later developments of combination CVRs would include the more potent estrogen found in combination OCs, ethinyl estradiol.
Norethindrone acetate, a progestin used in OCs, was paired with ethinyl estradiol in a core-design CVR. A CVR releasing 650 μg/day of norethindrone acetate and 30 μg/day of ethinyl estradiol was associated with serum levels of both norethindrone and ethinyl estradiol comparable with women ingesting an oral contraceptive containing 30-μg ethinyl estradiol and 1.5-mg norethindrone acetate.24,25 In a comparison of CVR users and OC users with these formulations, serum progesterone levels remained low, indicating inhibition of luteal activity in both groups. Side effects were reported with similar frequency by both groups, except nausea, which was greater for CVR users. The lipid profile of CVR users differed from OC users. CVR users had a significant increase in triglycerides and HDL levels. Other serum lipid levels including LDL and total cholesterol were unchanged with the use of this CVR.
Another formulation releasing 1 mg of norethindrone acetate per day and 20 μg of ethinyl estradiol per day was designed to be used cyclically for 1 year.26,27,28 This CVR had an outer diameter of 58 mm and inner diameter of 50.4 mm. This CVR was not associated with the same high rate of ovulation suppression as the previous formulation.26 Despite this, two large clinical trials demonstrated excellent contraceptive efficacy. There were no pregnancies in 654 women-months of use of this CVR in one study and one pregnancy in 831 women-months of this CVR use in another.26,28
This CVR was designed, like other combination CVRs, to be used in a cyclic fashion to provide regular withdrawal bleeding. Similar to previous trials of combination CVRS, most episodes of bleeding occurred during the scheduled withdrawal week. Episodes of intermenstrual bleeding (bleeding with the CVR in place) were uncommon, occurring in less than 2% of treatment days and less than 20% of treatment cycles.26 Furthermore, the breakthrough bleeding that did occur, usually began immediately before the time of scheduled CVR removal. Amenorrhea was rare. Nearly all subjects experienced a scheduled withdrawal bleed during each cycle. Other side effects and outcome parameters were similar to previous studies of combination CVRs. Body weight was not affected by this CVR. The changes observed in serum lipid levels were comparable with the trial of the earlier norethindrone combination CVR. After 1 year of CVR use, average serum levels increased for all lipid parameters. On average, serum triglycerides increased 20% to 33%, HDL increased 2% to 12%, total cholesterol 3% to 9% and LDL 1% to 17% at each study site. None of these changes was considered clinically significant. Despite the high efficacy, acceptable bleeding patterns, and safety of this CVR, it was not developed for clinical use.
The Population Council, which is responsible for the development of many of the CVR formulations and many other contraceptive methods available today, has studied a novel progestin, 17-acetoxy-16 methylene-19 norprogesterone (Nestorone). Nestorone is unusual because although it has pharmacologic activity similar to other progestins, it has no oral bioavailability. It does have good parenteral bioavailability. When absorbed through the vaginal epithelium, Nestorone demonstrated contraceptive efficacy and minimal androgenicity. When combined with ethinyl estradiol, a CVR with Nestorone was designed for monthly cyclic use to be used for a 3-month period. A CVR containing 60 mg of Nestorone and 122 mg of ethinyl estradiol was associated with complete ovulation suppression and no significant change in serum lipids in 72 women-months.29
Subsequently, a comparative trial of a core-design CVR was performed for several formulations. This core-design CVR had an outer diameter of 58 mm and inner diameter of 50.4 mm and was designed for cyclic use over a 3-month period. In the comparative trial, four formulations were evaluated; progestin-only formulations of Nestorone or levonorgestrel acetate each releasing 100 μg/day or each of these progestins combined with ethinyl estradiol releasing 30 μg/day.30 As occurred in trials of other progestin-only and combination CVR formulations, ovulation suppression was better and bleeding patterns were more predictable with the two combined formulations than the two progestin only formulations. The combined Nestorone/ethinyl estradiol CVR was associated with the most predictable bleeding pattern. The five women who received this CVR reported no episodes of breakthrough bleeding for the study duration, a total of 15 cycles. Mean total cholesterol levels increased for all CVR formulations, but no formulation was associated with a clinically significant increase. All Nestorone containing CVRs were associated with increased triglyceride levels. Levonorgestrel-containing formulations, in contrast, were associated with a decrease in triglyceride levels. Nestorone combination CVRs have promise as a safe, effective, and acceptable method of contraception. Phase III trials of a combined Nestorone ethinyl estradiol ring are currently planned.
One CVR has recently been marketed for contraceptive use in the United States. Under the trade name NuvaRing, the core-design CVR contains etonogestrel and ethinyl estradiol. Etonogestrel, or 3-keto-desogestrel, a gonane progestin, is the active metabolite of desogestrel. This progestin has demonstrated efficacy and safety in a single-rod subdermal implant system, Implanon. Etonogestrel, in this implant, does not have atherogenic or thrombotic properties in studies evaluating markers of each.31,32 The material of the CVR is ethylene-vinyl-acetate, which is a polymer that is different than polysiloxane but similar to polysiloxane in that it is nontoxic and nonirritating. This CVR measures 54 mm in outer diameter and 50 mm in inner diameter and releases 120 μg/day of etonogestrel and 15 μg/day of ethinyl estradiol.
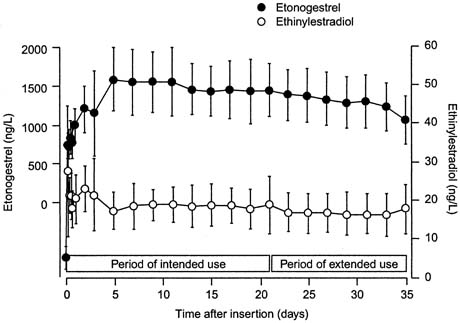
In a randomized trial of CVR users and OC users ingesting a combined oral contraceptive containing 150 μg of desogestrel and 30 μg of ethinyl estradiol, serum levels of 3-keto-desogestrel and ethinyl estradiol were compared.35 Similar to pharmacologic data with other CVR formulations, therapeutic levels of both hormones were reached by day 3 of use (Fig, 1). Also consistent with other CVR formulations, the peak serum levels of both hormones were lower in the CVR group compared with the OC group (Table 1). The peak serum levels of 3-keto-desogestrel in the CVR users were less than half those measured in combined oral contraceptive users, and peak serum levels of ethinyl estradiol were approximately one-fourth the levels of OC users. Also consistent with the pharmacokinetics of other ring formulations was the greater bioavailability (the area under the curve) of 3-keto-desogestrel (etonogestrel) in CVR users than combined OC users. The bioavailability of ethinyl estradiol was similar between the two groups.
|
|
| Etonogestrel/Ethinyl Estradiol CVR (N = 16) | Desogestrel/Ethinyl Estradiol Oral Contraceptive (N = 16) | |
| Etonogestrel | ||
| Peak serum concentration (ng/L) | 1716 ± 445 | 4273 ± 830 |
| Bioavailability (%) | 102.9 ± 12.8 | 79.2 ± 7.7 |
| Ethinyl estradiol | ||
| Peak serum concentration (ng/L) | 034.7 ± 17.5 | 124.9 ± 46.3 |
| Bioavailability (%) | 055.6 ± 12.9 | 053.8 ± 17.6 |
(Adapted from reference 35.)
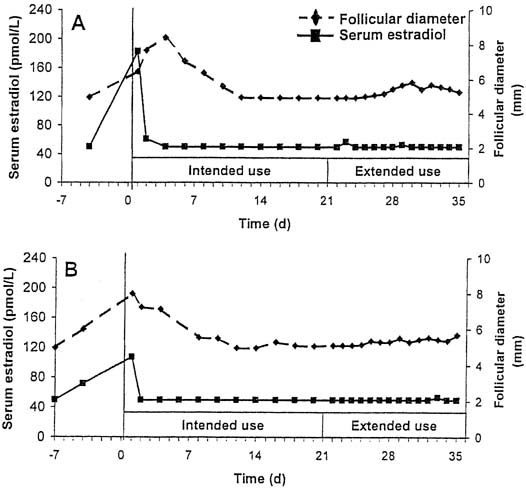
Although designed for cyclic use, with a withdrawal bleed in the fourth week after 3 weeks of use, this CVR provides serum levels of hormones that suppress ovulation for a period of up to 5 weeks. In a small study of extended use, subjects using the ring for a continuous 5-week period did not have a significant decrease in either serum etonogestrel or ethinyl estradiol levels.35 Ovulation inhibition was determined by measurement of follicular growth, serum progesterone, estradiol, and luteinizing hormone levels. Complete inhibition of ovulation was demonstrated with extended use of the CVR for up to 5 weeks (Fig, 2).36
|
A large, multicenter, international study demonstrated the safety and efficacy of this CVR.37 A total of 1145 women used this CVR for up to 13 cycles, for a total of 12,109 cycles observed. Only six pregnancies occurred in this study, yielding a failure rate of .65 per 100 women-years, confirming high efficacy of the CVR. Regular, scheduled withdrawal bleeding (during the ring-free week) was observed in almost all treatment cycles (97.9%–99.4%). A minority of subjects reported unscheduled bleeding or spotting with the CVR in place. Unscheduled bleeding or spotting was reported in only 2.6% to 6.4% of each of the 13 treatment cycles, most reporting irregular spotting (Table 2).
Table 2. Incidences (percentages) of Bleeding as a Proportion
of Intention-to-Treat Evaluable Cyclesa
| Irregular bleeding (breakthrough bleeding/spotting): | 2.6–6.4 |
| Withdrawal bleeding | |
| Absence | 0.6–2.1 |
| Earlyb | 5.4–7.7 |
| Early with spotting only | 2.8–5.4 |
| Latec | 20.4–27.3 |
| Late with spotting only | 16.5–21.4 |
aPercentages apply to cycles 1–12 for incidences of late withdrawal bleeding, and to cycles 1–13 otherwise.
bWithdrawal bleeding started while the CVR was in place.
cWithdrawal bleeding continued after the CVR was replaced.
(Reprinted from reference 37.)
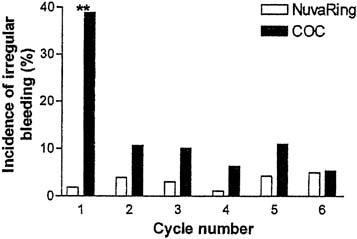
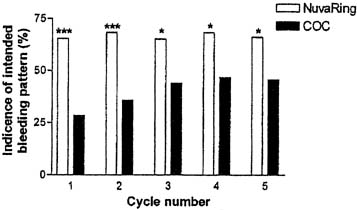
In a comparative trial of this CVR and an OC containing levonorgestrel 150 μg and ethinyl estradiol 30 μg, the CVR was equally effective and had a better bleeding pattern than the OC.38 There were no pregnancies during treatment among either the 126 OC users or the 121 CVR users. CVR users were more likely to experience regular withdrawal bleeding on schedule and less likely to experience irregular or unscheduled bleeding than OC users (Figs. 3 and 4). The incidence of hormonal side effects were also similar between the CVR and OC users, without a significant difference between them (Table 3). All treatment-related hormonal side effects were reported by less than 10% of subjects, with decreased libido (8.3%), nausea (5.0%), breast tenderness (4.1%), and vaginitis (4.1%) most commonly reported by CVR users. In addition, cervical cytology was not significantly different after treatment for either group.
|
|
| NuvaRing | COC Users | |
| AE | N (%) | N (%) |
| Acne | 02 (1.7) | 3 (2.4) |
| Breast tenderness | 05 (4.1) | 5 (4.0) |
| Decreased libido | 10 (8.3) | 0 (0.0) |
| Depression | 00 (0.0) | 6 (4.8) |
| Device-related events* | 03 (2.5) | NA (.0) |
| Headache | 04 (3.3) | 3 (2.4) |
| Leukorrhea | 03 (2.5) | 0 (0.0) |
| Nausea | 06 (5.0) | 4 (3.2) |
| Nervousness | 03 (2.5) | 2 (1.6) |
| Weight increase | 04 (3.3) | 2 (1.6) |
| Vaginal discomfort | 03 (2.5) | 0 (0.0) |
| Vaginitis | 05 (4.1) | 2 (1.6) |
NA, not applicable
*Comprising foreign body sensation, coital problems, and expulsion.
(Reprinted from reference 38.)
The Food and Drug Administration approved this CVR releasing 120 μg/day of etonogestrel and 15 mcg/day of ethinyl estradiol for use as a contraceptive in 2001. This CVR became available for use in 2002 with the trade name Nuvaring. The addition of a CVR to the already available reversible forms of contraception has greatly expanded the contraceptive choices for women in the United States.