Background
Medical abortion is relatively new technology. In Europe and China, a clinically useful medical alternative to surgical abortion has been present since 1988. Mifepristone in combination with a prostaglandin analogue has been used in France, Sweden, the United Kingdom, and China by more than 3 million women for abortion up to 63 days’ gestation. Beginning in late 1998, many other European countries approved mifepristone for sale and use, followed in September of 2001 by the United States. In the remainder of the world, these medications are not available because of political and economic restrictions on access to mifepristone. As such, alternative regimens have been developed that appear to have comparable overall efficacy.
History
Although the idea of using medications to induce a late menses or cause abortion dates back centuries, medically proven regimens have only been found in the last 50 years. In 1950, the folic acid antagonist 4-aminopteroylglutamic acid (Aminopterin) was shown to induce embryonic demise and resorption in mice and rats during the first week of gestation.3 The same investigators later used oral Aminopterin to induce medically indicated abortions in women less than 3 months’ gestation.4 Ten of 12 women aborted including all 6 patients less than 8 weeks’ and 4 of 6 at 8 or more weeks’ gestation. However, no further work in this area continued for many years.
In the 1970s, natural prostaglandins, such as prostaglandin E2 (PGE2) and prostaglandin F2α (PGF2α) were found to be effective at inducing abortion early in pregnancy when administered vaginally.5 Regimens that resulted in high efficacy caused intolerable nausea, vomiting, diarrhea, fever, and pain, necessitating premedication with analgesic, sedative, antiemetic, and antidiarrheal medications. Vaginal administration of effective doses of natural prostaglandins was also associated with intolerable levels of gastrointestinal side effects.6
Prostaglandin analogues were developed in the mid-1970s with more selective action on the myometrium so that lower doses could be used to effect abortion. These analogues, however, still had high rates of gastrointestinal side effects and were often unstable for long-term storage. By 1980, more stable analogues were found to be effective for abortion; gastrointestinal side effects were not as severe but still occurred with high frequency.7,8 Administration of gemeprost (16, 16-dimethyl trans Δ2 PGE1 methyl ester) in a 1-mg vaginal suppository every 3 hours for up to 5 doses resulted in complete abortion in 87% to 97% women up to 56 days’ gestation.8,9,10 However, 20% to 43% and 30% to 33% of subjects had vomiting and diarrhea, respectively.
An injectable analogue, sulprostone (16-phenoxy-tetranor PGE2 sulfonylamide) was also effective. In women up to 49 days’ gestation, 0.5 mg of sulprostone intramuscularly every 3 hours for 3 doses was as effective as suction aspiration (91% versus 94%, respectively).11 However, vomiting (30%) and diarrhea (21%) were still frequent with sulprostone. Attempts to lower the dose of sulprostone resulted in decreased efficacy.12
Thus, research with prostaglandin analogues had established these compounds as an effective alternative to surgical aspiration for early abortion. However, investigators reported, at that time, that the level of gastrointestinal side effects still limited the clinical utility of these agents.
While investigating compounds that would block glucocorticoid receptors, étienne-émile Baulieu noticed that some of the compounds bound strongly to the similarly shaped progesterone receptor and blocked the action of progesterone. Further refinement led to the production of mifepristone (RU-486), which began clinical testing in 1982. By 1988, mifepristone was licensed in France for use in combination with a prostaglandin analogue for early abortion.
In the United States, laboratory and clinical researchers studied mifepristone until 1989 when its import into the United States was banned. Because mifepristone was no longer available, even under research protocols, investigators searched for alternative medications to provide a medical abortion. With the publication of studies demonstrating the effectiveness of low-dose methotrexate for extrauterine pregnancy,13 this agent appeared to be a potential alternative. Research started in 1993 into the use of low-dose methotrexate and misoprostol for early abortion.
Interestingly, the consideration that prostaglandin analogues, when used alone, caused intolerable side effects began to be questioned by researchers as medical abortion became more prevalent in parts of the world. Consequently, renewed interest in prostaglandin analogues, specifically misoprostol, generated further studies of these agents as alternative medical abortion regimens.
Today, the most commonly used regimen throughout the world is mifepristone followed by a prostaglandin analogue, usually misoprostol. However, in areas without access to mifepristone, methotrexate and misoprostol and misoprostol alone are acceptable alternatives. Regardless of the regimen, it is truly the prostaglandin analogue that is causing the abortion. The actions of mifepristone or methotrexate “pretreatment” permits the use of lower doses of prostaglandin analogues as compared to when these agents are used alone.
Agents Used Currently for Medical Abortion
Background.
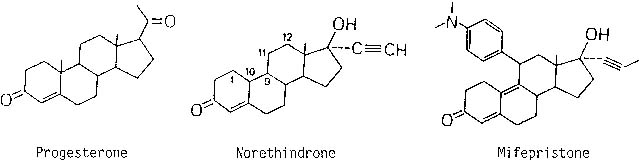
Progesterone is fundamentally important for sustaining an early pregnancy. Without progesterone support, the embryo is expelled by a prostaglandin-mediated mechanism. Clinically, inhibition of progesterone effects can be accomplished by preventing its synthesis or blocking its action at the receptor. Epostane, a 3β-hydroxysteroid dehydrogenase inhibitor, prevents synthesis of progesterone but requires dosing every 6 hours for 7 days to effect abortion.14,15 Mifepristone, a derivative of norethindrone, binds to the progesterone receptor with an affinity equal to progesterone itself16 but does not activate the receptor, thereby acting as an antiprogestin. To create mifepristone from norethindrone, a long side-chain is added at the 17-carbon position, which makes it bind tightly to the progesterone receptor, and a bulky side-chain is added at the 11-carbon position rendering it inactive (Fig. 1).
|
Mifepristone effects the uterus and cervix during early human pregnancy with specific actions that make it a potentially useful agent for abortion. First, mifepristone alters the endometrium by directly effecting the capillary endothelial cells of the decidua; there is no direct effect on the trophoblast.17 This results in separation of the trophoblast from the decidua causing bleeding and a decrease in human chorionic gonadotropin (hCG) secretion into the maternal circulation. The decidual action also increases prostaglandin release in vitro18 and in vivo.19 Second, mifepristone softens the cervix to allow expulsion.
Initial studies of mifepristone attempted to configure the optimal dose and dosing schedule to achieve acceptable rates of expulsion. The outcome of oral therapy was no different within a dose range of 50 to 400 mg daily in single or divided doses over 4 days. For gestations up to 49 days, complete abortion occurred in approximately 60% to 80%, incomplete abortion in 6% to 30%, and continuing pregnancies in 7% to 40%.15,20,21,22,23,24 At best, 80% of women treated with mifepristone alone in early pregnancy completely aborted, a rate not clinically acceptable. A U.S. study using a single large dose of 600 mg in women through 49 days’ gestation did demonstrate a complete abortion rate of 100% within 14 days for women with a serum β-hCG less than 5000 mIU/mL and 86% for higher serum β-hCG levels.25
In 1985, investigators reported that adding small doses of a prostaglandin analogue on the last day of mifepristone treatment increased the complete abortion rate to almost 100%.22,23 Ideally, these lower doses of prostaglandin analogue would also cause fewer side effects.
There are numerous other medical conditions for which mifepristone has been found to be important including emergency contraception, cervical ripening and labor induction, symptomatic leiomyomata uteri, endometriosis, Cushing’s syndrome, breast cancer, and glaucoma. Initial case series suggested that mifepristone would not be an effective treatment for extrauterine pregnancy26; these findings are plausible given the lack of progesterone receptor expression in fallopian tubes containing an ectopic pregnancy.27 However, recent reports suggest reasonable potential efficacy comparable to methotrexate28,29,30; obviously, more studies are needed.
Pharmacokinetics.
Mifepristone is easily absorbed when administered orally to humans; approximately 85% is absorbed but only 30% to 60% is bioavailable after first-pass metabolism.31 Peak serum concentration in pregnant and nonpregnant women is reached within 2 hours regardless of dose.32 Mifepristone has three major metabolites; all bind to the progesterone receptor, albeit much more weakly than mifepristone itself. Ovulation inhibition studies demonstrate that the threshold of mifepristone to begin to disturb the hypothalamic-pituitary axis is approximately 2 mg.33,34
The pharmacokinetics of mifepristone are different for daily doses less than 100 mg; at a dose of 100 mg or more, serum levels are similar.35,36,37 Over the first 72 hours, total serum concentration is similar for women administered 200 mg or 600 mg in a single dose.32 Similar peak serum concentrations of 2.0 to 2.5 μg/mL are found in women given 100 mg, 400 mg, 600 mg, and 800 mg of mifepristone.38 The nonlinear pharmacokinetics may be explained by the saturation of a specific transport protein for mifepristone, serum α1-acid glycoprotein; this protein is saturated at doses of 100 mg or more.39 The half-life of mifepristone is approximately 24 to 29 hours.22,36,38 Because of this long half-life, circulating levels remain significant at 48 hours.40 This information indicates that single doses of less than 600 mg are likely to be equally efficacious to 600 mg. Moreover, in women who received a single 600-mg dose of mifepristone, no difference in serum levels between those who did and did not abort was found for mifepristone or its metabolites.41
METHOTREXATE.
Background.
Methotrexate blocks dihydrofolate reductase, an enzyme necessary for the production of thymidine, a nucleotide necessary for DNA synthesis. Because the most rapidly dividing cells in the body need to make the most DNA, methotrexate will primarily affect these cells first. Medical conditions in which rapid cell division occurs include cancers, autoimmune diseases, and pregnancy. As such, methotrexate has been used for more than 40 years for treatment of neoplastic diseases, rheumatoid arthritis, and psoriasis. Other common uses include the treatment of systemic lupus erythematosus, dermatomyositis, severe asthma, Crohn’s disease, extrauterine pregnancy, and early abortion.
When used in the treatment of cancer, methotrexate is given in various types of high-dose regimens. Other medical conditions for which methotrexate can be used involve low doses; this is important because toxicity is dose dependent. High-dose versus low-dose, however, is not actually a comparison of absolute quantity administered at one time but the quantity administered over a given period of time (area under the curve).42 Thus, a patient who receives a single 85-mg injection of methotrexate is receiving low-dose treatment compared to a person who receives a 50-mg injection 5 days in a row, which is a relatively high-dose treatment.
Because methotrexate affects the most rapidly dividing cells of an unwanted medical condition, it can also affect rapidly dividing normal cells in the body. This accounts for the side effects seen with methotrexate use. Specifically, high-dose methotrexate can affect the lining of the gastrointestinal tract, bone marrow, and pulmonary interstitium. With high doses, renal toxicity and alopecia can also occur. However, with low doses such as those used for abortion, side effects are usually limited to minimal gastrointestinal problems such as nausea, vomiting, or diarrhea. In the literature, there have been only isolated reports of bone marrow suppression resulting in severe leukopenia,43 alopecia,44 and interstitial pneumonitis45 in patients treated with low-dose methotrexate for ectopic pregnancy.
Methotrexate is cytotoxic to neoplastic trophoblastic tissue (molar pregnancy and choriocarcinoma) and does not affect the ovary or oocytes. Gonadal dysfunction after high-dose combination chemotherapy treatment with regimens containing methotrexate has been reported but is felt to be unlikely to the methotrexate and more likely the result of alkylating agents such as cyclophosphamide.46 Women treated with chemotherapy including methotrexate for gestational trophoblastic tumors have normal future reproductive function.47 When methotrexate is used in a similar low-dose treatment for ectopic pregnancy, follow-up studies have demonstrated that menses returns normally and pregnancy rates are similar to those achieved by traditional surgical treatment of ectopic pregnancy.48 Finally, follow-up studies of women who received methotrexate for medical abortion suggest no impairment of fertility in the first year after the abortion.49 Thus, methotrexate has no effect on future fertility and does not increase the risk of anomalies in future pregnancies. Although fertility is unaffected, no long-term studies have addressed the theoretical possibility of decreased ovarian reserve and the potential impact on the age of menopause. However, a short-term low-dose treatment is unlikely to cause significant oocyte depletion.
Pharmacokinetics.
Limited pharmacokinetic information is available for methotrexate in pregnant women. Because of its potential for teratogenesis if used in early pregnancy, pharmacokinetic studies have typically involved men and nonpregnant women. Such studies have suggested that, at 24 hours after high-dose therapy, a methotrexate concentration of 5.0 μmol/L is indicative of an increased risk of toxicity.50
A single study has investigated methotrexate pharmacokinetics in early intrauterine pregnancy.42 In 10 women up to 49 days’ gestation who received 50 mg/m2 of methotrexate intramuscularly, serum levels peak within 1 to 2 hours, which is similar to that reported for nonpregnant subjects.51 The mean peak serum concentration was 4.4 ± 0.9 μmol/L and did not exceed 5.0 μmol/L at 24 hours in any patients. Serum levels were nondetectable within 48 hours. The mean area under the curve and renal clearance of methotrexate were 11.5 ± 2.2 μmol/hr per liter and 7.89 ± 1.98 L/hr, respectively. The renal clearance rate was the same as that for men and nonpregnant women receiving methotrexate in lower doses for rheumatoid arthritis or asthma.52,53,54
PROSTAGLANDIN ANALOGUES.
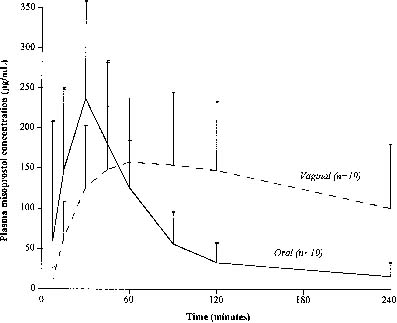
Gemeprost, sulprostone, and most other prostaglandin analogues available throughout the world require refrigeration, an important characteristic when considering their use in developing countries. Misoprostol ([±]-methyl-11α, 16-dihydroxy-16-methyl-9-oxoprost-13E-en-1-oate) is an inexpensive PGE1 analogue oral preparation that is stable at room temperature. Early favorable experience with mifepristone and misoprostol from France55 and the United Kingdom56 demonstrated that misoprostol increases uterine activity in early pregnancy. Pharmacokinetic evaluation of oral and vaginal misoprostol demonstrates that oral misoprostol is absorbed more rapidly, resulting in a higher peak serum level.57 However, the area under the curve following vaginal misoprostol is greater which may explain an improved effect (Fig. 2). Additionally, it is possible that there are direct cervical and uterine effects from vaginal administration.
|
Medical Abortion Treatment
Standard Regimen.
Mifepristone is typically used in clinical practice as a single 600-mg oral dose followed by a prostaglandin analogue. Analogues initially used with mifepristone included 1 mg of gemeprost vaginally or 0.25 mg of sulprostone intramuscularly. Abortion resulted in 95% to 96% of cases in women with pregnancies up to 49 days’ gestation.58,59 The single largest trial included 16,369 women up to 49 days’ gestation who received mifepristone with varying doses of gemeprost and sulprostone.59 Three hundred centers enrolled 16,369 patients, and 15,709 women were included in the final analysis. Of the patients excluded from the final analysis, 21 (0.13%) had an ectopic pregnancy. The overall efficacy was 95.3% (95% confidence interval [CI] 95.0%–95.6%) with no difference in treatment success rates by dose or type of prostaglandin. A small percentage (2.8%) of patients aborted after receiving mifepristone and before administration of the prostaglandin analogue. Within 4 and 24 hours after the prostaglandin analogue, 57% and 87%, respectively, had aborted. Failures included continuing pregnancies (1.2%), incomplete abortions (2.8%), and curettage because of heavy vaginal bleeding (0.7%). Hemorrhage requiring transfusion occurred in 11 (0.1%) cases. Vaginal bleeding lasted 8.3 ± 4.2 days but ranged from 1 to 60 days. Four serious cardiovascular complications (one myocardial infarction and three cases of severe hypotension) were associated with sulprostone injection. Consequently, the use of sulprostone in regimens with mifepristone for abortion has been abandoned.
Regimens of 600 mg of mifepristone and 1 mg of gemeprost vaginally were felt to be effective but large studies still demonstrated relatively high rates of vomiting (approximately 25%)60,61 and diarrhea (approximately 15%).60 Thus, researchers investigated the efficacy of lower doses of gemeprost in order to improve the side-effect profile. When 600 mg of mifepristone was followed 48 hours later by 0.5 mg of gemeprost, complete abortion occurred in 378 of 391 (96.7%, 95% CI 94.9%–98.5%) women up to 63 days’ gestation with no difference in efficacy according to gestational age.62 Nausea and vomiting were reported by 33% and 12% of subjects, respectively, within 2 hours of gemeprost administration, and diarrhea occurred in 7%. Thus, although not in a direct comparison to 1 mg of gemeprost, a dose of 0.5 mg appeared equally effective for pregnancies up to 63 days’ gestation but with slightly lower rates of gastrointestinal side effects.
The first large-scale study using the mifepristone/ misoprostol combination involved 873 French women up to 49 days’ gestation.63 The investigators described two consecutive studies in which all women received 600 mg of mifepristone orally followed approximately 48 hours later by 400 μg of misoprostol orally. Women in the second study were also eligible to receive an additional 200 μg of misoprostol orally if abortion had not occurred within 4 hours. Overall, 4% of women aborted solely from the mifepristone. Complete abortion occurred in 96.9% (95% CI 94.1%–97.7%) of the 488 women treated in the first study and in 98.7% (95% CI 96.8%–99.5%) of the 385 subjects in the second study, a nonsignificant difference. The abortion rates in the first 4 hours after misoprostol administration were 61% and 69% in the two studies, respectively, implying that the difference in overall efficacy was not necessarily a factor of the second dose. Uterine bleeding averaged 9 to 10 days, and one woman required a transfusion. Approximately 80% of women reported cramping for which about 15% received a nonopiate analgesic. After taking the misoprostol, 40% of the women experienced nausea, 15% vomiting, and 10% diarrhea. Thus, in women up to 49 days’ gestation treated with mifepristone, the use of misoprostol appeared equal in efficacy to gemeprost with similar side effects.
Two other large trials have evaluated the use of 600 mg of mifepristone orally followed approximately 48 hours later by 400 μg of misoprostol orally.64,65 A French group performed a multicenter trial including 1108 women up to 63 days’ gestation who were treated with this regimen but were also offered an additional 200 μg of misoprostol orally.64 Complete abortion rates were highest at lower gestational age: 97.6% up to 42 days’ gestation, 94.8% from 42 to 49 days’ gestation, 93.4% from 50 to 56 days’ gestation, and 86.8% from 57 to 63 days’ gestation. Similarly, continuing pregnancy rates increased with advancing gestational age: 0.8%, 1.4%, 1.6%, and 5.1%, respectively. Overall, 61.6% of women had not expelled the pregnancy within 3 hours of misoprostol administration and received a second dose. The group of women through 49 days’ gestation was compared with a historic cohort of women who received only a single dose of misoprostol. In this subgroup, there was no significant improvement in the expulsion rate as compared to the historic control during the initial monitoring after misoprostol administration (69.7% versus 64.9%, respectively), expulsion within 72 hours (92.7% versus 90.4%, respectively) and overall abortion rate (95.5% versus 95.4%, respectively).
The largest clinical trial combining 600 mg of mifepristone and 400 μg of misoprostol orally was the U.S. Population Council study.65 All participants had a vaginal ultrasound examination to confirm gestational dating; otherwise, the protocol was similar to that used in common practice in France. Patients received 600 mg of mifepristone orally, and returned 2 days later to receive misoprostol. If bleeding had occurred that might have been consistent with abortion (i.e., menses-like or heavier), an ultrasound examination was performed before misoprostol administration. Approximately 25% of patients had bleeding that met this criteria. Those women who had not aborted received 400 μg of misoprostol orally and were observed in the clinic or office for a minimum of 4 hours. Patients were then sent home and returned 12 days later for a final visit at which a physical or ultrasound examination was performed to confirm complete expulsion.
The overall effectiveness in the 2015 women evaluated was significantly worse using this treatment regimen for women with gestations more than 49 days (Table 1). The time of expulsion was known for 1468 subjects; 50% aborted within 4 hours and 84% within 24 hours after misoprostol administration. Vaginal bleeding lasted 17 ± 11 days; 9% of women had bleeding lasting more than 30 days. Four women received blood transfusions; one subject each through 49 days and at 57 to 63 days’ gestation and two subjects at 50 to 56 days’ gestation.
TABLE 1. Outcome of Treatment with Mifepristone (600 mg) Followed by Misoprostol (400 μg) orally for Abortion in the U.S. Population Council
Clinical Trial
Days’ gestation | ≤ 49 | 50–56 | 57–63 | all (≤ 63) |
Number |
827 |
678 |
510 |
2015 |
Mifepristone alone |
4.8% |
1.8% |
0.8% |
2.8% |
Total success |
92.1% |
83.0%* |
77.5%† |
85.4% |
Ongoing pregnancy |
1.0% |
3.7% |
9.0% |
3.9% |
Incomplete abortion |
4.7% |
7.5% |
7.1% |
6.3% |
Medical intervention‡ |
1.6% |
3.8% |
4.1% |
3.0% |
Patient request |
0.6% |
1.9% |
2.4% |
1.5% |
Total failure |
7.9% |
17.0% |
22.5% |
14.6% |
*Statistically different (p < 0.05) than group ≤ 49 days’gestation.
†statistically different (p < 0.05) than group ≤ 49 days’ and 50–56 days’ gestation.
‡Hemorrhage.
Adapted from Spitz IM, Bardin CW, Benton L, et al: Early pregnancy termination with mifepristone and misoprostol in the United States. N Engl J Med 338:1241, 1998.
Alternative Regimens.
Despite the early acceptance of the standard regimen of mifepristone 600 mg followed 48 hours later by a prostaglandin analogue in the clinic or office in women up to 49 days’ gestation in France and most other countries,66 investigators questioned whether alternative regimens using these agents would cause fewer side effects, be less expensive, or be more acceptable to providers and patients. The innovations studied have involved lower doses of mifepristone, the use of misoprostol vaginally, home administration of the misoprostol, and altering the timing of the misoprostol dose.
Lower Doses of Mifepristone.
As early as 1993, randomized trials had demonstrated equal efficacy 200 mg, 400 mg, and 600 mg of mifepristone when followed 48 hours later by 1 mg of gemeprost vaginally.61 Such results would be expected given the pharmacokinetics of mifepristone. Two randomized trials have evaluated lower doses of mifepristone in combination with misoprostol.
The first trial included 220 women treated with 200 mg or 600 mg of mifepristone (110 subjects per group) followed by 600 μg of misoprostol orally 48 hours later.67 There was no difference in clinical outcome, efficacy, bleeding, or pain between different mifepristone treatment groups. The overall effectiveness was 97.5% at up to 49 days’ gestation, 91.3% at 50 to 56 days’, and 84.4% at 57 to 63 days’. Although this trial showed equal efficacy and side effects when a dose of 600 μg of misoprostol was used, it did not address the more commonly used regimen of 600 mg of mifepristone and 400 μg of misoprostol; theoretically, the 400-μg regimen may have equal efficacy with fewer side effects.
The second trial was performed by the World Health Organization68 and included women up to 63 days’ gestation who received either 200 mg (n = 792) or 600 mg (n = 797) of mifepristone followed 48 hours later by 400 μg of misoprostol orally. Complete abortion rates were similar for both groups (89% and 88%, respectively) and, as in the U.S. Population Council study,65 were gestational-age–dependent: up to 42 days, 8%; 43 to 49 days, 11%; 50 to 56 days, 13%; and more than 56 days, 20%. Side effects were also not different between the groups. Thus, this study clearly demonstrated the clinical equality of 200 mg and 600 mg of mifepristone. Because mifepristone is the more expensive of the medications, such a regimen is more economical.
In addition to these randomized trials, three large studies using 200 mg of mifepristone combined with 800 μg of misoprostol vaginally in more than 4000 women have also confirmed the efficacy of the lower dose of mifepristone.69,70,71
Still, based on the pharmacokinetics, a dose of 100 mg would be theoretically effective. Creinin and colleagues72 reported a small trial in which all women received 100 mg of mifepristone and then were randomized to use misoprostol 48 hours later in a dose of 400 μg orally (n = 40) or 800 μg vaginally (n = 40). Women returned for follow-up evaluations 24 ± 1 hours after using the misoprostol and then 2 to 3 weeks later. If abortion had still not occurred and the pregnancy was nonviable, the subject returned again after an additional 3 weeks. At 24 hours after receiving misoprostol, 34 of 40 (85%, 95% CI 71%–94%) women who used oral misoprostol and 38 of 40 (95%, 95% CI 85%–99%) women who used vaginal misoprostol had complete abortions. Overall, complete abortion without surgical intervention occurred in 34 of 40 (85%, 95% CI 71%–94%) women and 40 of 40 (100%, 95% CI 91%–100%) women, respectively (p = .03). Side effects occurred with similar frequency in both treatment groups.
A World Health Organization-sponsored trial evaluated the clinical use of even lower doses of mifepristone combined with gemeprost in 1224 women.73 Participants were randomized to one of four groups using two different mifepristone doses (50 mg and 200 mg) and two different gemeprost doses (0.5 mg and 1.0 mg). Complete abortion rates were 85% and 90% in the 50-mg mifepristone groups using 0.5 mg and 1.0 mg of gemeprost, respectively. In the 200-mg mifepristone group, complete abortion rates were 92% and 93%, respectively. Importantly, a continuing pregnancy was four to eight times more likely with the lower mifepristone dose, regardless of the gemeprost dose. Overall, after adjustment, the relative risk of failure was 1.6 for the lower mifepristone dose.
Vaginal Misoprostol.
As the use of misoprostol with mifepristone was becoming more common, reports began to appear in the literature (for second-trimester abortion and in combination with methotrexate for early abortion) about using misoprostol vaginally with fewer gastrointestinal side effects. El-Rafaey and colleagues74 first reported the use of vaginal misoprostol in combination with mifepristone in 1994. Of 100 women up to 63 days’ gestation, 99 completely aborted within 4 hours of misoprostol administration and 67% did not require any pain medication. El-Rafaey and colleagues75 followed this study with a randomized trial in women up to 63 days’ gestation comparing 600 mg of mifepristone followed by 800 μg of misoprostol orally or vaginally (130 and 133 subjects, respectively, per group) 48 hours later. Slightly less than 3% of patients aborted from just the mifepristone, 95% aborted with the vaginal misoprostol and 87% with oral misoprostol. Moreover, 93% of the vaginal misoprostol group aborted within 4 hours compared to 78% of the oral group. The incidence of nausea (60% versus 70%), vomiting (31% versus 44%), and diarrhea (18% versus 36%) was much lower with the vaginal compared to the oral misoprostol group. However, one would expect the oral group to have much higher rates of gastrointestinal side effects than previously reported because a dose of 800 μg rather than 400 μg was used. Importantly, this randomized trial confirms that the use of vaginal misoprostol allows the effective use of mifepristone regimens through 63 days’ gestation (whereas oral misoprostol is only substantially effective through 49 days’ gestation), induces faster expulsion, and results in a lower rate of ongoing pregnancy.
Moreover, as discussed previously, three large studies have reported the clinical efficacy of regimens using 800 μg of misoprostol vaginally. Ashok and colleagues69 reported a retrospective evaluation of 2000 women who received 200 mg of mifepristone followed 36 to 48 hours later by 800 μg of misoprostol vaginally. In 928 women up to 49 days’ gestation and 1072 women from 50 to 63 days’ gestation, complete abortion occurred in 98.5% (95% CI 97.7%–99.3%) and 96.7% (95% CI 95.7%–97.8%), respectively. Continuing pregnancy occurred in only 0.2% (95% CI 0%–0.5%) and 0.8% (95% CI 0.2%–1.3%), respectively. Schaff and colleagues70,71 published two reports evaluating a 200-mg dose of mifepristone followed 48 hours later by 800 μg of vaginal misoprostol. In the first trial, which included 933 women with pregnancies up to 56 days’ gestation, complete abortion occurred in 97% and 96% of women up to 49 days (n = 660) and 50 to 56 days’ gestation (n = 273), respectively.70 The rates of continuing pregnancy were only 0.3% and 1.1%, respectively. In the second trial, which included 1237 women with pregnancies up to 63 days’ gestation, complete abortion occurred in 98%, 97%, and 96% of women up to 49 days’ gestation (n =578), 50 to 56 days” gestation (n =251), and 57 to 63 days’ gestation (n = 308), respectively.71 As in the first trial, the rates of continuing pregnancy were similar and extremely low (0.2%, 0.4%, and 1.0%, respectively).
Home Administration of Misoprostol.
Schaff and colleagues76 found home administration of vaginal misoprostol after mifepristone in pregnancies up to 56 days’ gestation to be safe and effective. Subjects had an option of returning 2 days later for vaginal administration of misoprostol 800 μg or inserting the tablets themselves at home. Participants were also instructed to return before misoprostol administration if bleeding had occurred after taking mifepristone that was equivalent to or heavier than menses. The mean gestational age was 43 days. Of the 166 subjects, 22 (13.3%) had significant bleeding after mifepristone requiring evaluation. Eight women (4.8%) aborted just with mifepristone; because this is almost double the 2.5% to 3% typically reported with this dose of mifepristone,59,63,75 it most likely represents that the cohort included many women at a very early gestational age. Only 3 (1.9%) of the remaining 158 women requested that the clinician place the misoprostol. All but 3 subjects (98.2%) aborted without surgical intervention. These 3 subjects had an aspiration for incomplete abortion. Nine (5.4%) found home administration unacceptable. This study again finds a high success rate with vaginal misoprostol administration and suggests that home administration may be safe and acceptable.
Multiple investigators have continued to publish reports of successful use of self-administered misoprostol in mifepristone medical abortion regimens.70,71,72,77,78,79,80,81 In the three largest trials using 200 mg of mifepristone and 800 μg of misoprostol vaginally,70,71,77 90% of subjects in all studies found home use of misoprostol acceptable regardless of prior abortion experience,70 gestational age,71 or time between mifepristone and misoprostol use.77 A total of 4 (0.1%) participants in two studies70,71 experienced adverse events in the hours after misoprostol administration. Two of these women presented for an emergent aspiration for heavy bleeding; neither required a blood transfusion. One woman had a syncopal episode while bleeding and fell and broke her nose. One patient had a vasovagal reaction to cramping and was treated with intravenous fluids. Only the latter occurrence (1 of approximately 4500 women) would have necessarily been avoided with in-office observation.
Time Interval Between Mifepristone and Misoprostol.
Based on the initial clinical trials using mifepristone and a prostaglandin analogue, the standard time to administer the prostaglandin analogue after mifepristone is 36 to 48 hours. However, regimens with a shorter interval between mifepristone and misoprostol administration, if effective, would lessen the amount of time necessary for a medical abortion to occur and, potentially, increase acceptability. Additionally, because approximately half of women bleed during the 48 hours after mifepristone administration,65,70 administering the drugs on the same day would decrease such an undesirable side effect. There are no studies that identify the time interval at which the increased reactivity to prostaglandin analogues occurs in a pregnant uterus.
Creinin and colleagues78 investigated earlier administration of the misoprostol using the standard doses of mifepristone and misoprostol in a randomized trial. Eighty-six women up to 49 days’ gestation received 600 mg of mifepristone followed on the same day (group 1) or 2 days later (group 2) by 400 μg of misoprostol orally. Women who received misoprostol on the same day as the mifepristone received a second misoprostol dose at the standard 48-hour interval if expulsion had not occurred. Complete abortion at 24 hours after the misoprostol occurred in 21 of 42 (50%, 95% CI 35%–65%) women in group 1 and 40 of 44 (91%, 95% CI 82%–99%) women in group 2 (p < .0001).
Schaff and colleagues77 performed a multicenter randomized trial in 2295 women up to 56 days’ gestation who self-administered 800 μg of misoprostol vaginally 24, 48 or 72 hours after taking 200 mg of mifepristone orally. Follow-up occurred within 8 days after the mifepristone. The misoprostol dose was repeated if vaginal ultrasound examination did not confirm expulsion. Complete medical abortion occurred in 98% (95% CI 97%–99%), 98% (95% CI 97%–99%), and 96% (95% CI 95%–97%), respectively. Additionally, the time waiting for expulsion was acceptable in 86%, 79%, and 76%, respectively (p = .001).
The investigators followed this study with a randomized trial to compare oral and vaginal dosing of misoprostol 24 hours after administration of 200 mg of mifepristone.79 Women up to 63 days’ gestation were randomly assigned to use either 2 doses of oral misoprostol, 400 μg, taken 2 hours apart or misoprostol, 800 μg, vaginally. Women returned for follow-up within 5 days and received vaginal misoprostol if a continuing pregnancy was present. Of the 1144 women who complied with their random assignment, complete abortion by the first follow-up visit occurred in 90% and 97%, respectively (p = .001). By the second follow-up visit, the complete abortion rates were 95% and 99%, respectively (p = .001). There were minimal differences in side effects. Despite the lower efficacy, women preferred the oral route.
More recently, Pymar and colleagues80 and Fox and colleagues81 reported that using mifepristone 200 mg and misoprostol 800 μg vaginally 6 to 8 hours apart caused expulsion within 24 hours in approximately 90% of women through 63 days’ gestation. Further investigations are ongoing to compare efficacy, side effects and acceptability of same day administration of the medications to administration 24 hours apart.
Comparative Trials with Different Agents.
Randomized clinical trials have been performed comparing vaginal and oral misoprostol following mifepristone up to 49 days’ gestation82 and vaginal misoprostol and gemeprost following mifepristone treatment through 49 days’ gestation.83
Aubeny and Chatellier82 treated 237 women through 49 days’ gestation with 600 mg of mifepristone and then randomly assigned the women to receive 400 μg of misoprostol orally (n = 119) or vaginally (n = 118). If expulsion had not occurred within 3 hours, a second dose of misoprostol was administered. Only 1 participant required suction aspiration for an overall abortion rate of 99.6% (95% CI 97.3%–100%). The abortion rates between the oral and vaginal misoprostol groups were equal at 3 hours (71.0% versus 68.3%) and 1 hour after the repeat misoprostol dose (76.9% versus 77.0%). There were no differences in side effects or bleeding patterns. Acceptability questionnaires suggested a preference in both groups for oral administration. This study demonstrates a high success rate, even for oral misoprostol, through 49 days’ gestation. The results contradict larger trials that demonstrate lower success rates through 49 days’ gestation and a study by the same researchers with a repeat dose of misoprostol which demonstrated a 3-hour expulsion rate of 48% and a success rate of 95.5%. This study shows equal efficacy between oral and vaginal misoprostol administration when the dose is repeated in 3 hours but does not provide information about whether providing a repeat dose is superior to a single dose. Obviously, a regimen that does not require repeat dosing is simpler for patients to use.
The same researchers also submitted a letter to the editor stating their success rates with the oral misoprostol regimen with repeat dosing at 3 hours when indicated in 1973 women through 49 days’ gestation.84 The success rate within 3 hours was 68.1% (95% CI 66.1%–70.2%) and the overall success rate was 98.4% (95% CI 97.9%–99.0%). Although this information suggests a high success rate with repeat dosing of the misoprostol, it does not prove superiority over single dosing with an oral or a vaginal regimen. Interestingly, Schaff and colleagues79 found only a 90% expulsion rate when using mifepristone 200 mg followed 24 hours later by 400 μg of misoprostol orally with a repeat dose in all subjects. Thus, there still are no randomized trials that prove the benefit of repeat administration of misoprostol within a few hours of the initial dose.
Bartley and colleagues83 performed a multicenter randomized trial comparing misoprostol and gemeprost approximately 48 hours after 200 mg of mifepristone in women up to 63 days’ gestation. The doses of misoprostol (800 μg vaginally) and gemeprost (0.5 mg vaginally) have been well established as highly effective in combination with mifepristone for medical abortion. A blinded observer observed all 996 women for 4 to 6 hours after prostaglandin analogue administration. A follow-up visit occurred approximately 2 weeks later with a clinician blinded to the treatment group; ultrasonography was performed to confirm expulsion only if the clinician felt one was clinically indicated. Eighty-six (8.6%) women did not follow-up and no information was obtainable from a referring physician. Expulsion during the observation period occurred in 69.6% and 69.3% of women who received misoprostol and gemeprost, respectively. The overall complete abortion rate was significantly higher after treatment with misoprostol than gemeprost (98.7% versus 96.2%, respectively; p = .02). Importantly, continuing pregnancy was much less common with misoprostol than gemeprost (0.2% versus 1.8%, respectively; p = .02). In both groups, failure was more common at higher gestational ages. Side effects were not clinically significantly different between groups. Thus, this trial demonstrates that misoprostol, when administered vaginally, should be the prostaglandin analogue of choice for use with mifepristone for medical abortion.
Clinical Use of Mifepristone Regimens.
Current published research supports the use of mifepristone orally in a dose of 200 mg or 600 mg. Clinical trials demonstrate no difference in efficacy between the two dosages; thus, the lower dose should be used because it is less expensive. Moreover, pharmacokinetic studies of serum levels show no significant difference at any dose of 100 mg or greater. All Rh-negative patients should receive Rh-immune globulin when the mifepristone is ingested, although the necessity of Rh-immune globulin for medical abortion patients has been questioned.85
Mifepristone and misoprostol regimens have higher efficacy through 42 days’ gestation compared to 43 to 49 days’ gestation.64,68,86 The upper limit of gestational age acceptable for using a mifepristone regimen for abortion is dependent on the type and route of administration of the prostaglandin analogue. Regimens using 0.5 mg of vaginal gemeprost or 800 μg of misoprostol are effective through 63 days’ gestation, although a randomized trial suggests that the misoprostol provides superior efficacy.83 When 400 μg of oral misoprostol orally is used with mifepristone, the regimen is clinically effective through 49 days’ gestation. Finally, when the dose of mifepristone is decreased to 200 mg and combined with misoprostol 600 μg orally, the regimen is effective through 56 days’ gestation although a 49 days’ gestation limit may be more appropriate.67
The medical evidence strongly supports that after mifepristone administration, the patient can safely and effectively administer misoprostol herself, either orally or vaginally. The misoprostol, when administered vaginally, can be used between 24 and 72 hours after the mifepristone with equal efficacy; however, patients tend to prefer earlier administration. A follow-up evaluation is typically performed 10 to 15 days after misoprostol administration to ensure that there are no complications. Expulsion can be confirmed by clinical history or ultrasonography. Typically, European, Chinese, and U.S. clinical trial protocols have included a surgical aspiration if the abortion is not complete by this follow-up visit. Schaff and colleagues76 have demonstrated that this intervention is not necessary and additional doses of misoprostol or simply waiting (if the pregnancy is not viable) is an acceptable alternative.
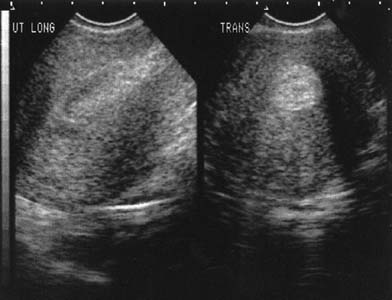
Interestingly, earlier follow-up can be performed if vaginal ultrasonography is used to confirm expulsion. Clinicians must understand what is a normal transvaginal view of the uterine cavity after misoprostol administration, which will contain obvious clot and, sometimes, fluid (Fig. 3). The main goal of the ultrasound examination is solely the determination of the presence or absence of the gestational sac. Harwood and colleagues87 demonstrated that the mean endometrial thickness 24 hours after using misoprostol in women with a complete medical abortion was 17.5 mm but ranged from 7.6 to 29.0 mm. Typically, clinicians define a thickness greater than 15 mm as suspicious for incomplete abortion. One week after the abortion, the mean thickness was 11.3 mm but ranged from 1.6 to 24.9 mm. Fifteen percent of women had an endometrial lining of 16 mm or greater and, traditionally, would be diagnosed as an incomplete abortion. Because all of the women in this trial had a complete abortion, this study points out that it is normal to visualize clot and debris in the uterus when transvaginal ultrasound is used to evaluate a patient after medical abortion. Vaginal bleeding after a medical abortion using mifepristone varies among studies but, surprisingly, the variation is not due to use of different doses of either medication or the route of administration of the misoprostol. Three studies, including two from France, found an average duration of bleeding of 9 days,63,64,75 with a range of 1 to 32 days.63,75 However, the remainder of studies, including those from the United States, report a mean duration of bleeding of 14 to 17 days67,70,76 with a range of 1 to 69 days.65,76,88
|
Davis and colleagues89 specifically followed women with bleeding diaries with the intent of ascertaining accurate bleeding patterns. Mifepristone, 200 mg, and misoprostol, 800 μg vaginally, were administered as part of a larger trial comparing misoprostol administration 1, 2, or 3 days after the mifepristone.77 Of the 138 women enrolled for the bleeding analysis, 78% returned their diaries in person or by mail. Subjects reported bleeding for a mean of 14 days and spotting for a mean of 10 days. Overall, women had bleeding or spotting for an average of 24 days, much longer than what is typically in efficacy studies. Twenty percent of women had bleeding or spotting that lasted more than 35 days. The results were not effected by when the misoprostol was administered. However, gestational age was directly related to the length of bleeding, with a mean total number of days of bleeding or spotting of 19 days in gestations less than 6 weeks and 25 days for equal to or greater than 6 weeks. Additionally, there was no significant difference in bleeding duration based on contraceptive method started after expulsion. Two interesting studies from China have also demonstrated that it is safe to offer women combined hormonal contraception one day after misoprostol adminstration.90,91 Use of oral contraceptives did not effect abortion outcome, duration of vaginal bleeding, or blood loss as collected on sanitary pads.
METHOTREXATE AND MISOPROSTOL.
Regimens.
Clinical trials of methotrexate and misoprostol for early abortion began 11 years after human testing on mifepristone had begun. Initial studies using methotrexate 50 mg/m2 intramuscularly followed 3 days later by misoprostol demonstrated the vaginal use of misoprostol was more effective than oral administration.92
Two larger trials were conducted suggesting potential benefits of a longer interval between methotrexate and misoprostol administration. Hausknecht93 reported 178 women who received 50 mg/m2 of methotrexate intramuscularly followed 5 to 7 days later by 800 μg of misoprostol vaginally in women up to 63 days’ gestation. Patients received a repeat misoprostol dose if the gestational sac was not expelled. A 96% success rate by 14 days after methotrexate administration was reported; 14% of these patients had required a repeat misoprostol dose. However, 26% of patients were less than 35 days’ gestation and 66% were less than 45 days. Additionally, no follow-up past 1 to 2 weeks after the misoprostol was performed. Thus, incomplete expulsion may not have been properly identified. Also, only 2 of 178 women were reported to have methotrexate side effects, a rate notably lower than that reported in other series.
The second report was a randomized trial94 comparing the efficacy of 50 mg/m2 of methotrexate followed by 800 μg of vaginal misoprostol 3 and 7 days later in pregnancies up to 56 days’ gestation. The misoprostol dose was repeated if the gestational sac was not expelled. The overall effectiveness rates were 83% and 98% in the 3- and 7-day groups, respectively. A reanalysis of the data attributes the difference in overall effectiveness to gestational age.95 Whereas complete abortion rates up to 49 days’ gestation were 90% and 96% (p = .37) for the 3-day and 7-day groups, respectively, corresponding rates at 50 to 56 days’ gestation were 75% and 100% (p = .04). Only 65% and 68%, respectively, of patients in the two groups expelled the pregnancy within 24 hours of the first or second doses of misoprostol.
A multicenter trial was then performed including 300 women up to 56 days’ gestation who received 50 mg/m2 of methotrexate intramuscularly followed 7 days later by 800 μg of misoprostol vaginally.96 The misoprostol dose was repeated 24 hours later if abortion did not occur. Overall, 88% of subjects completely aborted without the need for a surgical procedure. The success rate was significantly better up to 49 days’ gestation (90.6%, 95% CI 85.7%–94.2%) compared to 50 to 56 days (81.6%, 95% CI 72.5%–88.7%, p = .04). Only 70% of subjects had completed the abortion by 14 days after the methotrexate (Table 2). Women who passed the pregnancy within 24 hours of the first or second dose of misoprostol (immediate success) experienced bleeding and spotting for a total of 14 ± 7 days. Those women who passed the pregnancy after a delay had bleeding and spotting for 11 ± 9 days. Side effects after injection of methotrexate were reported by 43.7% of subjects although some of the side effects could just be a result of morning sickness. After misoprostol administration, nausea occurred in 12%, vomiting in 8%, diarrhea in 7%, and subjective fever or chills in 3% of subjects.
TABLE 2. Cumulative Complete Abortion Rate after Treatment with Methotrexate
and Misoprostol up to 49 Days’ Gestation
Regimen with Methotrexate 50 mg/m2 IM66 (n = 300) | Regimen with Methotrexate 50 mg Orally68 (n = 299) | |
Before misoprostol |
1% |
0% |
After first dose of misoprostol* |
50% |
70% |
After second dose of misoprostol* |
64% |
78% |
By day 14 |
66% |
81% |
By day 21 |
73% |
83% |
By day 28 |
83% |
87% |
By day 35 |
87% |
89% |
By day 45 |
87% |
91% |
IM, intramuscularly.
* Within 24 hours of misoprostol administration.
Because of encouraging results of a preliminary evaluation of regimens using oral methotrexate,98 a multicenter trial was conducted using a 50-mg dose followed by misoprostol 800 μg vaginally.97 Overall, 91% of subjects completely aborted, with 78% expelling the pregnancy within 24 hours of the first or second doses of misoprostol (Table 2). For these women, bleeding and spotting lasted a total of 15 ± 8 days. Women who expelled the pregnancy after a delay had bleeding and spotting for a total of 11 ± 7 days. Side effects reported after oral methotrexate occurred more often than with intramuscular administration even though the relative amount of methotrexate was less; this difference might be a result of the route of administration, different patient populations, or the manner in which side effects data were reported. Misoprostol side effects were also reported more frequently than in previous reports including nausea in 33%, vomiting in 18%, diarrhea in 18%, dizziness in 18%, headache in 12%, and fever/chills in 31%.
Carbonell and colleagues99-101 performed multiple clinical studies of methotrexate and misoprostol using complicated treatment regimens in which all women were instructed to douche with boiled water the night prior to misoprostol administration and to dampen each misoprostol tablet with 2 to 3 drops of water prior to insertion. Subjects were also advised to lie down for 3 hours after misoprostol administration. Misoprostol was repeated every 48 hours up to three total doses. Additionally, once expulsion was documented by vaginal ultrasonography at follow-up examination, the subject used another 400 to 1200 μg of vaginal misoprostol over the next 24 hours. The investigators reported efficacy of 93% in 207 women up to 49 days’ gestation and 92% in 80 women from 50 to 63 days’ gestation who used 800 μg of misoprostol vaginally 3, 4, or 5 days after 50 mg/m2 of methotrexate intramuscularly (no difference in efficacy by day of misoprostol administration, p = .97).99 Another randomized trial by Carbonell and colleagues101 compared 25 and 50 mg of methotrexate orally followed 7 days later by 800 μg of misoprostol vaginally in 310 women up to 56 days’ gestation. Complete abortion rates were 91% and 90% (p = .93) in the 25- and 50-mg methotrexate groups, respectively, with no significant differences in side effects. The majority of treatment failures had continuing cardiac activity on ultrasound examination. Side effects after the methotrexate were similar between the groups and included nausea in 11% to 12%, vomiting in 5% to 6%, and diarrhea in 2% to 3%. Stomatitis was reported by 3% of women receiving 25 mg and 5% of women receiving 50 mg of oral methotrexate (p = .4). Side effects reported after misoprostol use included nausea in 30%, vomiting in 24%, diarrhea in 59%, and chills in 60%.
Because the results of Carbonell and colleagues99,100,101 were quite different from other authors, especially in regard to how quickly expulsion occurred and abortion rates at gestational ages beyond 49 days, Creinin and Carbonell collaborated in a randomized trial conducted in both of their research offices comparing 50 mg/m2 of methotrexate intramuscularly followed 5 to 6 days later by either moistened or dry 800 μg of misoprostol vaginally in 240 women up to 49 days’ gestation.102 The misoprostol was self-administered at home by the participants after quickly dipping each tablet into tap water before vaginal insertion using the technique as dictated by Carbonell. The misoprostol dose was repeated 7 days after the methotrexate if the abortion had not yet occurred. The repeat dose was administered by the clinician using a speculum. If the subject had been randomlt assigned to use moistened misoprostol, then the clinician also added 1 to 2 mL of tap water with the repeat misoprostol dose. The proportion of women who aborted within 24 hours of the initial or repeat dose of misoprostol did not differ between the moistened and dry misoprostol groups (84% versus 81%, respectively; p = .65), although a trend toward a higher rate with moistened misoprostol was evident at the U.S. site (87% versus 76%, p = .19, n = 124). The overall success rates by 35 days after methotrexate were also similar in the two misoprostol groups (95% versus 92%, respectively; p = .40). Interestingly, the success rates in the research office of Carbonell were much lower than reported previously.99,100,101 Subjects randomly assigned to the moistened misoprostol group reported more diarrhea (36% versus 21%; p < .05) and fever/warmth/chills (44% versus 30%; p < .05) than the dry misoprostol group. Thus, from this randomized trial, it appears that using moistened misoprostol in methotrexate medical abortion regimens does not improve complete abortion rates but does cause an increase in side effects.
Wiebe and colleagues103 performed an interesting trial involving 100 women up to 49 days’ gestation who were randomly assigned to receive 50 mg/m2 of the parenteral form of methotrexate intramuscularly or orally (in 10 mL of orange juice) followed by 600 μg of moistened misoprostol vaginally. The misoprostol was repeated if the woman experienced only light vaginal bleeding. The success rates for the oral and injected forms were 95% and 89%, respectively (p = .30). A sample size of 830 would have been required to establish a significant difference. When patients were given a choice between oral and intramuscular administration, 57% chose the oral route.
Comparative Trials with Different Agents.
A randomized trial in Canada compared the efficacy and side effects of 600 mg of mifepristone followed 36 to 48 hours later by 400 μg of misoprostol orally (n = 518) and 50 mg/m2 of methotrexate intramuscularly followed 4 to 6 days later by 800 μg of misoprostol vaginally (n = 524) in women up to 49 days’ gestation.104 All misoprostol doses were self-administered by the women and repeated 24 hours later if bleeding was less than typical menstrual flow. Subjects returned 7 days after the initiation of treatment at which time vaginal ultrasonography was performed; 800 μg of misoprostol was administered vaginally if the examination demonstrated persistence of a gestational sac. A suction aspiration was performed if a viable gestation was present at a second follow-up visit 2 weeks after initiation of treatment of if the gestational sac had not expelled by 5 weeks after initiation of treatment. The abortion rate by the 1-week follow-up examination was 75% in the methotrexate group and 90% in the mifepristone group (p < .001). The surgery rates were 4% in both groups (Table 3). Side effects were similar between the mifepristone/misoprostol and methotrexate/misoprostol regimens with statistically significant differences in the incidence of headache after the mifepristone or methotrexate (19.1% versus 11.3%, p = .001), diarrhea after the misoprostol (15.9% versus 27.0%, respectively; p < .001), fever after the misoprostol (11.5% versus 21.7%, respectively; p < .001), chills after the misoprostol (23.2% versus 49.3%, respectively; p < .001), and headache after the misoprostol (28.6% versus 17.0%, respectively; p < .001). The mean number of bleeding days was significantly greater with the mifepristone regimen (14.6 versus 13.3 days, p = .32) whereas the mean pain score using an 11-point scale was significantly greater with the methotrexate regimen (6.3 versus 5.8, p = .003).
TABLE 3. Complete Abortion Rate in a Prospective Randomized Trial Comparing
Mifepristone or Methotrexate Followed by Misoprostol up to 49 Days’ Gestation
Mifepristone Regimen* | Methotrexate Regimen† | |
Subjects randomized |
524 |
518 |
Subjects lost to follow-up |
1 |
2 |
Subjects without physical examination for follow-up |
7 |
4 |
Subjects available for analysis |
516 |
512 |
Complete abortion rates |
||
By 7 days after initiating treatment‡ |
91.9% |
75.4% |
After 7 days after initiating treatment† |
3.9% |
20.5% |
Medically indicated surgical aspiration |
3.3% |
2.0% |
Surgical aspiration by patient request |
1.0% |
2.1% |
*Mifepristone, 600 mg, followed 36 to 48 hours later by misoprostol, 400 μg, orally. Misoprostol dose was repeated 24 hours later if the subject experienced bleeding less than her usual period. If expulsion had not occurred by follow-up 7 days after the mifepristone, the subject received moistened misoprostol, 800 μg, vaginally.
†Methotrexate 50 mg/m2 IM followed 4 to 6 days later by moistened misoprostol, 800 μg, vaginally. Misoprostol dose was repeated 24 hours later if the subject experienced bleeding less than her usual period. If expulsion had not occurred by follow up 7 days after the mifepristone, the subject received moistened misoprostol, 800 μg, vaginally.
‡p < .001
Adapated from Wiebe E, Dunn S, Guilbert E, et al: Comparison of abortions induced by methotrexate or mifepristone followed by misoprostol. Obstet Gynecol 99:813, 2002.
Clinical Use of Methotrexate Regimens.
Methotrexate can be administered as either 50 mg/m2 intramuscularly or 50 mg orally in pregnancies up to 49 days’ gestation. Over 49 days, the efficacy appears to decrease.
Vaginal ultrasound has been used to confirm gestational age in all large trials performed using these agents. All Rh-negative patients should receive Rh-immune globulin; if the blood type is not known at the time methotrexate is administered, the patient can receive Rh-immune globulin anytime before she uses the misoprostol. The misoprostol dose (800 μg) can safely be self-administered by the woman at home using the same tablets that are used for oral dosing. The literature supports administration of the misoprostol 3 to 7 days after the methotrexate.
Follow-up is performed approximately 1 week after receiving the methotrexate at which time a vaginal ultrasound is performed. The gestational sac will be absent in approximately 70% to 80% of patients; in these women, treatment is complete. If the gestational sac is still present, the misoprostol dose is repeated either in the office by the clinician or a second dose is given to the woman to self-administer when she returns home. Approximately 25% of these patients will abort within 24 hours. If no gestational cardiac activity is present, the patient can return in 4 weeks. The patient returns in 1 week if embryonic cardiac activity is present. At that time, the vaginal ultrasound is repeated and a suction abortion performed if embryonic cardiac activity is still present. If the gestational sac is absent, treatment is complete. If the sac is still present but no embryonic cardiac activity present, the patient can return in 3 weeks for follow-up.
Almost all subjects who do not expel the pregnancy within 24 hours of the first or second dose of misoprostol will pass the pregnancy after a delay of a few days to a few weeks. On average, expulsion will occur approximately 3 to 3 1/2 weeks after methotrexate administration. During the time after misoprostol administration that the woman is waiting for the pregnancy to pass, her bleeding will stop and she will not feel pregnant. Most commonly, she will have slight cramping or light bleeding a day or two before the pregnancy passes. The patient can be reassured that she will not have heavy bleeding and severe cramping without any notice. Of the patients who have to wait for the pregnancy to pass, approximately 96% will expel the pregnancy completely within 35 days after the methotrexate injection.96
Vaginal bleeding and spotting after methotrexate and misoprostol last a similar length of time for immediate success patients, averaging 14 to 17 total days, with a range of 2 to 52 days.96,97,105,106 The duration of bleeding is shorter for delayed success patients after treatment with methotrexate and misoprostol, averaging 10 to 11 days with a range of 1 to 55 days.96,97,105,106 Even with relatively long periods of bleeding, significant changes in hemoglobin are very rare.
Although equally effective when compared to the standard regimen of mifepristone and misoprostol, regimens using methotrexate and misoprostol act much less rapidly.104 Methotrexate offers the advantage of being inexpensive; the drug cost of methotrexate for abortion is less than four U.S. dollars and misoprostol is less than $1.00.
MISOPROSTOL ALONE FOR MEDICAL ABORTION.
Regimens.
The first evaluation of misoprostol alone as an abortifacient suggested that the agent, even when used vaginally, was ineffective. Creinin and colleagues105 randomly assigned 61 women up to 56 days’ gestation to methotrexate, 50 mg/m2 intramuscularly, or no pretreatment followed 3 days later by misoprostol, 800 μg, vaginally. The misoprostol was repeated 24 hours later if abortion did not occur. Complete abortion rates were 90% and 47% (p < .001) with the methotrexate/misoprostol and misoprostol-only regimens, respectively. The investigators discontinued the study early because of the low efficacy of the misoprostol only regimen. Further studies in 1996107,108 involved more frequent and potentially inconvenient dosing using vaginal misoprostol, 200 to 400 μg every 4 to 12 hours. Approximately two thirds of women aborted within 48 hours of treatment. Similarly, özeren and colleagues109 reported a complete abortion rate of 58% in 36 women less than 63 days’ gestation (mean, 47 days) after 800 μg of misoprostol was administered vaginally. The misoprostol was repeated 3 days later if needed.
Despite this discouraging data for misoprostol alone, independent investigators reported using moistened misoprostol successfully as a single agent for abortion.
Carbonell and colleagues110,111,112,113,114,115 have published six studies evaluating the use of misoprostol alone for early first-trimester medical abortion. As with their studies using methotrexate and misoprostol, all participants were instructed to douche with boiled water the night prior to misoprostol administration and to dampen each misoprostol tablet with 2 to 3 drops of water prior to insertion. Subjects were also advised to lie down for 3 hours after misoprostol administration.
The first trial included 141 women through 69 days’ gestation.110 Subjects received 800 μg of misoprostol vaginally every 48 hours as needed for up to 3 doses. After expulsion was documented by vaginal ultrasound, the subject used another 400 μg to 1200 μg of vaginal misoprostol over the next 24 hours. Complete abortion occurred in 94% (95% CI 89%–98%); 83% aborted after the first dose of misoprostol. Complete abortion occurred in 107 of 111 (96%, 95% CI 9%–100%) women up to 63 days’ gestation compared to 25 of 30 (83%, 95% CI 70–97%) women 64 to 70 days’ gestation. At the time of the report, side effects were more frequent as compared to previously reported regimens including mifepristone or methotrexate. With this misoprostol regimen, nausea was reported by 24% of women, vomiting in 25%, diarrhea in 58%, dizziness by 20%, headache by 13%, warmth by 35%, and chills by 57%. These rates are similar to those reported with the use of other prostaglandin analogues in the past that were considered clinically unacceptable.
The investigators used an identical protocol in 175 women up to 63 days’ gestation.111 Complete abortion occurred in 92%. Success with a single dose of misoprostol occurred in 78%. Rates of side effects were relatively identical to those reported in their prior study.110 A third and larger study with 720 women up to 63 days’ gestation also used a similar protocol except for a shortened misoprostol dosing interval of 24 hours.112 The rates of complete abortion were 92%, 90%, and 87% for 35 to 49, 50 to 56, and 57 to 63 days’ gestation, respectively (p = .37). Two patients (0.3%) required blood transfusions after hemoglobin levels decreased to 6.1 and 5.7 g/dL. Rates of side effects were similar to the two prior studies.110,111
Carbonell and colleagues113 continued to pursue alternative dosing regimens in women up to 63 days’ gestation, performing one study in 90 women using 600 μg of misoprostol vaginally every 8 hours,113 another study in 150 adolescents using 800 μg of misoprostol vaginally every 24 hours,114 and a third study in 300 women using 1000 μg of misoprostol vaginally every 24 hours.115 In all three trials, the same method of misoprostol administration was used as previously described and up to 3 doses of misoprostol was administered to induce expulsion. The latter two trials had outcomes and side effect rates were similar to those previously published with other regimens. The regimen with 600 μg of misoprostol vaginally every 8 hours effected complete abortion in 64% (95% CI 53%–74%).113 Because this regimen was different than other regimens both in dose and interval of misoprostol administration, it is unclear if one or both factors are relevant to the efficacy of misoprostol alone for early abortion. None of these trials involved a randomized comparison to previously studied methods.
Jain and colleagues116,117,118 have reported three studies using moistened misoprostol with complete abortion rates similar to those reported by Carbonell and colleagues.110,111,112,113,114,115 The first study included 100 women up to 56 days’ gestation.116 The investigators placed the misoprostol in the vagina with a speculum and then moistened the tablets with 2 mL of normal saline. Subjects remained recumbent for 30 minutes. A second 800-μg vaginal misoprostol dose was given if the subjects did not abort within 24 hours. The overall rate of successful abortions was 88% using this regimen with 73% of women completing the abortion within 24 hours of a single dose of misoprostol, and the remainder of the subjects completing the abortion within 6 days of the second dose of misoprostol. The percentage of women who required analgesia was 79%, and the percentages of women who experienced vomiting, diarrhea, and fever/chills were 28%, 44%, and 68% respectively. These side effects occurred more frequently than a matched historical cohort of women who were given 600 mg of oral mifepristone followed by 400 μg of oral misoprostol (p < 0.001 for requiring analgesia, diarrhea and fever/chills, and p = .01 for vomiting).
Jain and colleagues117 then completed a randomized trial using 800 μg of wet misoprostol 48 hours after a 24-hour treatment with tamoxifen (n = 75) or placebo (n = 75) in women up to 56 days’ gestation. The vaginal misoprostol dose was repeated 24 hours and 8 days later if the abortion did not occur. Complete abortion rates were similar with or without tamoxifen (93% and 91%, respectively). Vomiting, diarrhea, and fever/chills were 45%, 67%, and 89% in the misoprostol-only group. Rates for the tamoxifen and misoprostol group were similar, suggesting no effect of the tamoxifen in effecting abortion. Given the high rates of side effects with moistened misoprostol, the investigators evaluated pretreatment with loperamide and acetaminophen as compared to a historic cohort who did not receive pre-treatment.118 One hundred subjects up to 56 days’ gestation received 800 μg of misoprostol vaginally, moistened with 2 mL of saline and was repeated every 24 hours for up to 3 doses. The complete abortion rates were not significantly different between the groups (93% versus 89%, respectively). The pretreated group used significantly less opiate analgesia and experienced less diarrhea (23% versus 44%, p = .003). However, the incidence of fever or chills and emesis was not improved with pretreatment.
Comparative Studies with Different Agents.
Jain and colleagues119 performed a randomized, double-blind trial in women up to 56 days’ gestation using 200 mg of mifepristone (n = 119) or placebo (n = 125) followed 48 hours later by 800 μg of misoprostol vaginally. Misoprostol was repeated every 24 hours up to three doses. All women received pretreatment before misoprostol with loperamide and acetaminophen. Complete abortion rates were 95.7% and 88.0%, respectively (p < 0.05). The women who received mifepristone aborted much more quickly and required fewer doses of misoprostol as compared to women who received misoprostol alone (Table 4). The complete abortion rates by gestational age did not reach statistical significance: 96.0% and 88.8% for the mifepristone and misoprostol versus placebo and misoprostol groups through 49 days’ gestation and 95.5% and 86.7%, respectively from 50 to 56 days’ gestation. Women experienced slightly more side effects with the mifepristone regimen but that may be because of a higher rate of treatment success resulting in effects related to the abortion process (Table 5).
TABLE 4. Complete Abortion Rate in a Prospective Randomized Trial Comparing
Mifepristone or Placebo Followed by Misoprostol up to 56 Days’ Gestation*
Mifepristone Regimen | Misoprostol Regimen | |
Subjects randomized |
125 |
125 |
Subjects without confirmation of misoprostol administration |
1 |
0 |
Subjects lost to follow-up |
5 |
0 |
Subjects available for analysis |
119 |
125 |
Complete abortion rates† |
||
By 24 hours after misoprostol |
89.9% |
72.0% |
Between 25 and 48 hours after misoprostol |
5.8% |
13.6% |
Between 49 and 72 hours after misoprostol |
0 |
2.4% |
Medically indicated surgical aspiration |
0 |
9.6% |
Surgical aspiration by patient request |
4.2% |
2.4% |
*Mifepristone, 200 mg, or placebo followed approximately 48 hours later by moistened misoprostol, 800 μg, vaginally. The misoprostol dose was repeated every 24 hours for up to 2 more doses if expulsion had not occurred.
†Cumulative abortion rates statistically different at 24 hours (p < .001), 48 hours (p = .012), and 72 hours (p = .047).
Adapated from Jain JK, Dutton C, Harwood B, et al: A prospective randomized, double-blinded, placebo-controlled trial comparing mifepristone and vaginal misoprostol to vaginal misoprostol alone for elective termination of early pregnancy. Hum Reprod 17:1477, 2002.
TABLE 5. Side Effects from Placebo, Mifepristone, and Misoprostol Treatment
in a Prospective Randomized Trial Comparing Mifepristone or Placebo
Followed by Misoprostol up to 56 Days’ Gestation*
Placebo and Misoprostol | ||
Side effects after mifepristone or placebo | ||
Nausea† |
57.0% |
40.5% |
Vomiting† |
32.7% |
12.9% |
Diarrhea |
7.6% |
7.0% |
Fever or chills |
27.1% |
19.0% |
Side effects after misoprostol | ||
Nausea |
43.9% |
51.7% |
Vomiting |
32.7% |
21.5% |
Diarrhea |
60.9% |
71.5% |
Fever or chills |
5.1% |
3.2% |
*Mifepristone, 200 mg, or placebo followed approximately 48 hours later by moistened misoprostol 800 μg, vaginally. The misoprostol dose was repeated every 24 hours for up to 2 more doses, if expulsion had not occurred.
†p < .01
Adapated from Jain JK, Dutton C, Harwood B, et al: A prospective randomized, double-blinded, placebo-controlled trial comparing mifepristone and vaginal misoprostol to vaginal misoprostol alone for elective termination of early pregnancy. Hum Reprod 17:1477, 2002.
Clinical Use of Misoprostol Regimens.
Misoprostol, 800 μg, vaginally, using a complex dosing regimen with repeat doses every 24 hours as indicated, is an effective method for medical abortion. The reported rates of side effects, though, are high, compared to methods that include pretreatment with mifepristone or methotrexate. Vaginal bleeding averages a total of 11 to 13 days,111,112,119 but no studies have reported the total range of vaginal bleeding. Use of acetaminophen pretreatment decreases the incidence of narcotic use and loperamide pretreatment decreases the incidence of diarrhea.
The expulsion rate after a single dose of misoprostol appears to be much lower than what occurs after mifepristone administration and equal to what occurs using dry misoprostol in a simple regimen following methotrexate administration (Table 6). This method, similar to methotrexate and misoprostol, is inferior in efficacy to a mifepristone/misoprostol regimen. However, misoprostol is inexpensive and widely available throughout the world. Thus, given the relatively limited availability of mifepristone throughout the world, misoprostol alone may be a reasonable option for women who want to avoid or do not have access to a suction abortion.
TABLE 6. Cumulative Abortion Rate with Moistened Vaginal Misoprostol as
a Single Agent for Medical Abortion
Cumulative Abortion Rate | |||||||||
Year | Number | Misoprostol Dose | Maximum Gestational Age | Interval (hours) Between Doses | Maximum Number of Doses | Additional Misoprostol* | First | Second | Third |
1997110 |
141 |
800 μg |
70 days |
48 |
3 |
yes |
83.0 |
92.9 |
93.6 |
1997111 |
175 |
800 μg |
63 days |
48 |
3 |
yes |
77.7 |
91.4 |
92.0 |
1999112 |
720 |
800 μg |
63 days |
24 |
3 |
yes |
65.4 |
83.5 |
89.6 |
1999116 |
100 |
800 μg |
56 days |
24 |
2 |
no |
73.0 |
88.0 |
— |
2000117 |
75 |
800 μg |
56 days |
24 and 144 |
3 |
no |
73.3 |
86.7 |
90.7 |
2000113 |
83 |
600 μg |
63 days |
8 |
3 |
yes |
38.6 |
55.4 |
62.7 |
2000114 |
150 |
800 μg |
63 days |
24 |
3 |
yes |
60.0 |
85.3 |
88.7 |
2001115 |
300 |
1000 μg |
63 days |
24 |
3 |
yes |
69.3 |
89.7 |
93.0 |
2002119 |
125 |
800 μg |
56 days |
24 |
3 |
no |
72.0 |
85.6 |
88.0 |
*Treatment after abortion documented with additional doses of misoprostol ranging from 400 μg to 1200 μg vaginally.
MEDICAL ABORTION IN THE LATE FIRST TRIMESTER.
The definition of medical abortion classically includes the use of medicines to effect abortion through 9 weeks’ gestation.1 A few clinical trials have evaluated mifepristone and misoprostol and misoprostol alone for medical abortion during the first trimester but after 9 weeks’ gestation. In England and Wales, nearly 75% of abortions are performed from 9 to 13 weeks’ gestation120 compared to approximately 33% in Scotland121 and the United States.122 Ashok and colleagues123,124 have performed two clinical trials evaluating the use of mifepristone and misoprostol in this gestational age range. The first trial included 66 nulligravidas and 54 multigravidas from 9 to 13 weeks’ gestation.123 All women received 200 mg of mifepristone followed 36 to 48 hours later by 800 μg of misoprostol vaginally administered in a clinic. The women received up to two additional doses of 400 μg of misoprostol orally or vaginally every 3 hours; the route was dependent on the amount of vaginal bleeding. Expulsion occurred on the day of misoprostol treatment in all women, although 5% required curettage for an incomplete abortion. Nearly 16% of women required antiemetics, 32% experienced diarrhea, and 22% required parenteral analgesia. Vaginal bleeding lasted an average of 12.5 days but ranged from 1 to 43 days.
Based on this initial experience, the same investigators performed a trial including 445 women between 10 and 13’ weeks gestation who were allowed to choose their method of abortion, either medical or surgical, or be randomized to a method if they did not have a strong preference for either.124 All women had an ultrasound examination to confirm gestational age. Medical abortion was performed using 200 mg of mifepristone followed 36 to 48 hours later by 800 μg of misoprostol vaginally administered in a clinic. The misoprostol was repeated every 3 hours for up to two more doses if abortion had not occurred using 400 μg of misoprostol either orally or vaginally depending on the amount of vaginal bleeding. If evacuation did not occur, a surgical evacuation was performed. All suction abortions were performed under general anesthesia.
Seventy-seven women had a method preference: 15 for medical abortion and 62 for surgical abortion. The remaining women were randomly assigned to medical (n = 188) or surgical (n = 180) abortion. There were no demographic differences between the subject populations. Of the 203 women who had a medical abortion, 94.6% expelled the pregnancy, although 105 of the subjects (51.7%) were less than or equal to 70 days’ gestation. A trend was present for decreasing efficacy with advancing gestational age but the numbers in each gestational week were too small to be statistically significant. The complete surgical abortion rate was 97.9%. The success rates were not statistically different overall and during any gestational week, albeit, the numbers were small. Women having a medical abortion experienced significantly more side effects whether they chose the method or were randomized to the method. The total days of bleeding were not significantly different for those women who chose their method but was statistically significantly greater in the women who were randomized to medical abortion as compared to surgical abortion (14.2 days versus 11.2 days, p < .0001). There was no statistical difference in pain evaluations. This study supports the potential use of medical abortion in women up to 10 weeks’ gestation and suggests that it could possibly be safely used on an outpatient basis in women up to 13 weeks’ gestation.
Carbonell and colleagues125,126,127 reported three studies including women beyond 9 weeks’ gestation using moistened misoprostol as a single agent in repeat doses. All of the studies used a complex method of administering the misoprostol with saline through a speculum and requiring the patient to remain recumbent after administration. As with other studies by the same investigators, additional misoprostol was administered after confirmation of pregnancy expulsion.
A study of 120 adult women125 and another study of 150 adolescent women127 both used 800 μg of misoprostol vaginally with repeat doses every 24 hours up to a maximum of three doses. The adult women had a gestational age range from 64 to 84 days and cumulative complete abortion rates after the first, second, and third misoprostol doses of 72.5%, 81.7%, and 86.7%. The adolescent women ranged from 63 to 84 days and abortion rates of 58.0%, 76.7%, and 84.0%, respectively. In both reports, the authors indicated that the side effects were of “low intensity and short duration.”
A third study by Carbonell and colleagues126 used a similar protocol in women from 64 to 91 days’ gestation except that the misoprostol dose was repeated every 12 hours for up to three doses. Seventy-two percent of subjects were from 63 to 76 days’ gestation and 93% were from 63 to 83 days’ gestation. Although there was no statistical difference in expulsion rates by gestational age, a trend was present suggested less success beyond 83 days’ gestation. Cumulative complete abortion rates after the first, second and third misoprostol doses were 63.9%, 77.2%, and 85.0%. All side effects other than pain and diarrhea were reported to have occurred by less than 30% of subjects, and all side effects other than pain disappeared within 2 hours. Because the majority of subjects were less than 77 days’ gestation, this study can only be heralded to provide accurate information about potential efficacy and side effect rates for women through 76 days’ gestation. Thus, it appears that a 12-hour dosing interval provides similar efficacy and side effects in this gestational age range compared to what the same investigators found using 24-hour dosing intervals.
In summary, these few studies suggest that medical abortion can safely be performed after 9 weeks gestation. The expulsion rates using misoprostol alone are inferior to those using a combination of mifepristone and misoprostol as demonstrated by Ashok and colleagues.124 Most importantly, more information is needed to understand if these agents can be used safely between 9 and 13 weeks’ gestation on an outpatient basis, and to compare safety, efficacy, and acceptability in larger trials with outpatient surgical abortion.
Providing Medical Abortion
GENERAL INFORMATION.
Patient counseling must first stress early pregnancy options to be sure that a woman is certain about her decision to have an abortion. A medical abortion is not a means to make abortion an easier decision for a woman uncertain about whether to continue the pregnancy. The woman must decide herself if she wants a medical abortion; the medical team should not make the decision for her. If she is uncertain, then the decision about abortion technique must be delayed until she has reached a firm decision, even if the delay means that she will be unable to choose a medical option.
Once a woman is sure that she desires an abortion, counseling can then focus on the methodological options; this discussion needs to include the risks, benefits, and side effects of both medical and surgical abortion. It is important to emphasize the necessity of follow-up and number of visits for a medical abortion. Bleeding will be much heavier than with menses (potentially accompanied by severe cramping) and is best described to patients as comparable to a miscarriage. Even more important is to emphasize that medical abortion, rgardless of the regimen, does not work as well as early surgical abortion, which has a failure rate (reaspiration) of less than 1%.128 Finally, the counselor and physician still must comply with all local and federal regulations.
Medical contraindications to abortion with mifepristone include confirmed or suspected ectopic pregnancy, intrauterine device in situ, long-term systemic corticosteroid administration, adrenal failure, known coagulopathy, allergy to mifepristone, and inherited porphyria. Methotrexate is contraindicated in women with an allergy to methotrexate, known coagulopathy, active liver or renal disease, and an intrauterine device in situ. Asthma is not a contraindication because methotrexate is used in the treatment of severe asthma and misoprostol is a weak bronchodilator.129 Mifepristone has not been demonstrated clinically to affect asthma, but patients with severe asthma may require long-term corticosteroid use, which contraindicates mifepristone use. Misoprostol should be avoided in women with an allergy to misoprostol, uncontrolled seizure disorder, and acute inflammatory bowel disease. Additionally, because of a lack of data regarding the safety of these agents in women with significant medical conditions, special considerations should be used before providing medical abortion to women with severe anemia, severe liver, renal, or respiratory disease, uncontrolled hypertension, and cardiovascular disease (angina, valvular disease, arrhythmia, or cardiac failure).
The use of misoprostol during the first trimester for medical abortion, either with mifepristone, methotrexate, or by itself, has rarely been evaluated in women with a prior cesarean section. Only a single study has evaluated this issue. Xu and colleagues130 reported their findings from a cohort analysis of 192 women up to 49 days’ gestation who received 25 mg of mifepristone twice daily for 3 days and 600 μg of misoprostol orally on the fourth day. Thirty-five of these women had had a prior cesarean section and the remainder had no known uterine scar. Complete abortion rates were 94% (95% CI 81%–99%) and 90% (95% CI 75%–91%), respectively. No women in the group with prior cesarean section experienced any identifiable problems with their uterine scar. The duration of bleeding was similar between groups.
Although medical contraindications are infrequent, social or psychological contraindications to medical abortion are more common. Women are not good candidates for medical abortion if they do not wish to participate in their abortion or take responsibility for their care, are anxious to have the abortion over quickly, cannot return for follow-up visits, or cannot understand the instructions because of language or comprehension barriers. Other nonmedical criteria to be considered are access to a phone in case of an emergency, and distance from emergency medical treatment (i.e., suction curettage for hemorrhage).
Even though heavy bleeding is expected, it is helpful to identify for the patient how much bleeding is considered too much. An easy reference for the patient to use is soaking of 2 pads per hour for 2 hours in a row.131 This is not a point at which intervention is necessary, but a time when it is good to check in with the care provider. Whether or not it is imperative for the patient to seek emergency care is dependent at that point on how the patient is feeling, her baseline hemoglobin, if the bleeding seems to be slowing, and how far she is from emergency treatment. It may be helpful for the clinician to talk to the patient at 20- to 30-minute intervals until the bleeding slows as long as she is feeling all right; of course, this is dependent on the factors stated above.
Excessive bleeding is rare when medical abortion is restricted to pregnancies less than 49 menstrual days. The mean duration of bleeding is less than 2 weeks, but some women bleed until their next menstrual period. Because medical abortion is not 100% effective, 24-hour availability of surgical abortion services for cases of hemorrhage because of retained products of conception is a necessity. This is no different than what is required when patients are primarily treated by surgical abortion. Clinicians other than obstetrician–gynecologists who wish to provide medical abortion services must work in conjunction with an obstetrician–gynecologist or be trained in surgical abortion in order to offer medical abortion treatment.
Patients are supplied with appropriate instructions and oral narcotic analgesics. Patients should use ibuprofen or acetaminophen initially and only use oral narcotics if necessary. The use of a non-steroidal anti-inflammatory drug such as ibuprofen is not contraindicated and does not decrease the likelihood of abortion after prostaglandin analogue administration.132
CHOICE OF ABORTION METHOD.
Women who have decided to terminate their pregnancy tend to be comfortable with their choice of method, either medical or surgical, when they are given the opportunity to make their own choice.133 The most common reason a woman wants to have a medical abortion is because of a desire to avoid some aspect of a surgical procedure.133,134,135,136,137,138 Thus, one would expect acceptability studies to have positive results; because the study participants were actively seeking out an alternative to surgical abortion, they were very likely to be satisfied with their choice. Mifepristone regimens would be preferred for a next abortion in more than 70% of women who had a prior surgical abortion and by 80% to 96% of women overall.130,131,138,139,140 Additionally, mifepristone and misoprostol would be recommended to a friend by 95% of women in Vietnam136 and 96% of U.S. women.140 Similar results are found in studies using methotrexate and misoprostol.135,136,141,142,143,144
Randomized trials of acceptability are only helpful for counseling women who are truly unsure whether they prefer to have a medical or surgical abortion. Henshaw and colleagues133 randomized women with no method preference to the standard regimen of mifepristone and misoprostol or suction aspiration under general anesthesia. Almost all (98%) of the women who had a surgical abortion would recommend that method to a friend whereas, for medical abortion, the method was only acceptable to those women up to 49 days’ gestation. Because the standard regimen would not be used beyond 49 days’ gestation, this study only confirms that women with no preference tend to be comfortable with either method early in pregnancy.
Interestingly, Creinin145 randomized 50 women of 49 days’ gestation or less to medical and surgical abortion. The medical abortion regimen was 50 mg of methotrexate orally followed 5 to 6 days later with 800 μg of misoprostol vaginally. The misoprostol was self-administered at home and was repeated 1 to 2 days later if needed. The surgical abortion method was manual vacuum aspiration with local anesthesia in the office. Of the women randomly assigned to a surgical abortion, 92% (95% CI 81%–100%) stated they would choose a surgical for a next abortion, whereas only 63% (95% CI 43%–82%) of women randomly assigned to a medical abortion would choose that option in the future (p < .001).
TERATOGENICITY.
Congenital anomalies in continuing pregnancies that do not have a surgical abortion is a concern. There has been no evidence of a teratogenic effect of mifepristone. A 2-year study of 91,665 pregnancies in France revealed 3452 fetal malformations, however, none of the women who had malformed fetuses had taken mifepristone.146 Methotrexate is an antimetabolite that could damage a fetus; however, reports of teratogenicity involve high doses of methotrexate as used for chemotherapy or exceeding normal dose ranges.147,148 With low doses, reports are few, although a review of teratogenicity with low-dose oral methotrexate in early pregnancy found no effect.148
Misoprostol use in the first trimester is of greater teratogenic concern as it has been associated with two specific types of anomalies. Five cases of a frontal and/or temporal defect in the skull without other anomalies were described in women who had taken 400 to 600 μg of misoprostol orally and/or vaginally.149 Gonzalez and colleagues150 reported on seven cases of limb abnormalities, four of whom also had a diagnosis of Möbius sequence (mask-like faces with bilateral sixth and seventh nerve palsy and frequently coincident micrognathia). The mothers all had taken 200 to 1800 μg of misoprostol orally or vaginally between 4 to 12 weeks amenorrhea to attempt abortion. The authors suggested that these anomalies may represent a vascular disruption defect. There were three other children born with similar anomalies after maternal misoprostol ingestion. Two children had limb deficiencies (one with Möbius sequence) and one child had Möbius sequence; the gestational ages at the time of ingestion were not reported. Because misoprostol is the common agent used with both mifepristone and misoprostol, the potential for teratogenicity is an important issue for both types of medical abortion regimens. Wiebe141 reported six cases of limb reduction abnormalities in fetuses examined after failed abortion with methotrexate and misoprostol. These findings are consistent with the teratogenicity of misoprostol in medical abortion regimens. Schuler and colleagues151 published a prospective analysis of 86 infants exposed to misoprostol and 86 infants not exposed to misoprostol and found no significant difference in the rates of major or minor anomalies. However, they reported a significantly increased risk of spontaneous abortion after misoprostol exposure (17% versus 6%, respectively [relative risk, 3.0, 95% CI 1.1, 7.9]). This study is severely limited by small sample size.
As such, it must be emphasized to the patient before she begins a medical abortion that the agents being used to effect the abortion can cause anomalies in the ongoing pregnancy. Thus, she must be certain of her decision to have an abortion and be willing to have a surgical abortion should the medications not cause expulsion and the pregnancy still be viable.